Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H
Question:
Order each of the following sets of compounds with respect to SN2reactivity:
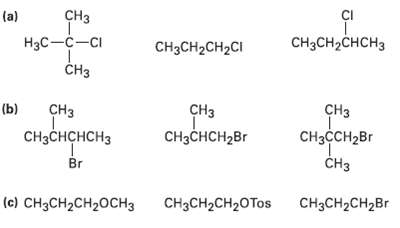
Transcribed Image Text:
CI CHз (a) CH3CH2CHCH3 Hас —с—сі CH3CH2CH2CI CHз (b) CHз CHз CHз CHзCнCH2Br CнзснсHсHз CнзссH2Br Br CHз (c) CH3CH2CH20CH3 CHяCH2CH2Br CHзCH2CH20Tos
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
SN2 reactivity Least reactive a b c CH3 HCCCl CH3 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a) i CH3CH2CHCH3 CI CH (b) (CH)3CI (CH)Br (CH)3C (c) Br CHCH3 C-Br CBr 3
-
Order each of the following sets from the least exothermic electron affinity to the most. a. F, Cl, Br, I b. N, O, F
-
Order the compounds in each of the following sets with respect to increasing acidity: (a) Acetic acid, oxalic acid, formic acid (b) p-Bromobenzoic acid, p-nitro benzoic acid, 2, 4-dinitrobenzoic acid...
-
Roberts Originals Co. (ROC) provides new and unique cases and otherassignments to professors each semester to ensure that students will not be able to find the solutions published online. Due to the...
-
Suppose you are an HR managers at a company with high turnover among middle managers (that is, many of them quit to work elsewhere.) Write a brief argument telling the company's executive why a...
-
Test for exactness. If exact, solve. If not, use an integrating factor as given or obtained by inspection or by the theorems in the text. Also, if an initial condition is given, find the...
-
In order for an employee performance appraisal system to work effectively, which of the following appraisal criteria should not be present? A. Criteria should be job related. B. Criteria should be as...
-
Document for Analysis: Information Request Your Task. Analyze the following poorly written letter, and list its weaknesses. If your instructor directs, revise it using the suggestions you learned in...
-
Ex 3-20 P.75 (10) An engineering from operates a job casting system. Production of is absorbed at the rate of 3.50 per machine hour. In order to allow for non-production on cost and profit, mark-up...
-
The capital investment for a new highway paving machine is $950,000. The estimated annual expense, in year zero dollars, is $92,600. This expense is estimated to increase at the rate of 5.7% per...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane: (a) CN (b) CH3CO2 (c) CH3S
-
List the qualities you would like to see in your mentor. Who do you knowin your immediate environment, from your educational program, in your specialty organization, in your local community, through...
-
Complete Exercises 2-B and 2-H in Writing and Analysis in the Law using what you learned in the reading and in the Seminar. Use paragraph form, use complete sentences, and make sure you use proper...
-
What is the value of a stock expected to be in 9 years if the annual dividend is expected to remain unchanged forever at $3.65, the expected rate of return is 6.9% per year, and the next dividend is...
-
Once invested IN a corporation, shareholders want their money out - they want a return on investment! John owns 2 5 % of REFUND CORP INC, which paid out a $ 5 0 , 0 0 0 distribution to him on 1 2 / 3...
-
Worksheet Financial Statement Ratios. Lowe's Companies, Inc Jan 28, 2022 and Jan. 29, 2021 Current Ratio Current Assets / Current Liabilities Acid Test Current Assets Current Liabilities (Cash + ST...
-
3. Peter Senen operates in a JIT manufacturing system. For August, Peter Senen purchased 10,000 units of raw materials at P1.00 per unit on account.What is the The journal entry to record the...
-
describe the main users of accounts and explain why they need accounting statements;
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
Consider the reaction: A solution is made containing an initial [Fe 3 + ] of 1.0 * 10 -3 M and an initial [SCN ] of 8.0 * 10 -4 M. At equilibrium, [FeSCN 2 + ] = 1.7 * 10 -4 M. Calculate the value...
-
Predict the products of saponification of the following esters. (a) (b) (c) (d) CH CHC-OCH-CH
-
Show how you would accomplish the following syntheses in good yields. (a) (b) (c) (d) (e) (f) (g) (h) NH-C-H NH2 COOH C-O-C CH HO COOH COCH(CH3)2 COOH COOH CHO CH2OH CHO COOH COOCH3 CH2OCCH3 COOCH...
-
Propose mechanisms for the following reactions. (a) (b) (c) (d) (e) (f) (g) Does this reaction proceed with retention, inversion, or racemization of the asymmetric carbon atom? Ph-CCI(CH3)2CHOHPhC...
-
A contractor constructed a house for resale, which was sold immediately. For tax purposes, this is an example of A) capital income. B) business income. C) other income. D) property income.
-
You invest $100 in a risky asset with an expected rate of return of 0.12 and a standard deviation of 0.15 and a T-bill with a rate of return of 0.05. What percentages of your money must be invested...
-
Nanometrics, Inc., has a beta of 3.43. If the market return is expected to be 13.50 percent and the risk-free rate is 7.00 percent, what is Nanometrics required return? (Round your answer to 2...

Study smarter with the SolutionInn App


