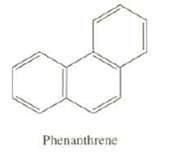
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond
Question:
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond of Phenanthrene would your predict to be the shortest?
Transcribed Image Text:
Phenanthrene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The five resonance structures are quite similar and are expected to m...View the full answer

Answered By

Jinah Patricia Padilla
Had an experience as an external auditor in Ernst & Young Philippines and currently a Corporate Accountant in a consultancy company providing manpower to a 5-star hotel in Makati, Philippines, Makati Diamond Residences
5.00+
120+ Reviews
150+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
Draw four reasonable resonance structures for the PO3F2- ion. The central P atom is bonded to the three O atoms and to the F atom. Show formal charges.
-
Draw contributing resonance structures for each of the following species, and rank the structures in order of decreasing contribution to the hybrid: a. b. c. d. e. f. CH3C-CH CHCH3 CH3 0 CH3COCH3 +OH...
-
You are the assistant vice president in charge of production for a firm that produces computers. Your firm's production function is f(L,K) = min (L,K) Where L and K are the quantities of the two...
-
Explain why it is important that the activities used to develop a flexible overhead budget are accurate cost drivers?
-
Compute the standard deviation of the payoffs for each decision. What does this tell you about the risk in making the decision? Slaggert Systems is considering becoming certified to the ISO 9000...
-
Water flows in a 5 -ft-wide rectangular channel with a flowrate of \(Q=30 \mathrm{ft}^{3} / \mathrm{s}\) and an upstream depth of \(y_{1}=2.5 \mathrm{ft}\) as is shown in Fig. P10.15. Determine the...
-
Ollie Mace is the controller of SDC, an automotive parts manufacturing firm. Its four major operating divisions are heat treating, extruding, small parts stamping, and machining. Last years sales...
-
Sara was a TV host under contract to Program A. Sara's contract with Program A contained negative covenants to the effect that she would not act as a presenter for anyone else. She breached her...
-
UPS prides itself on having up-to-date information on the processing and current location of each shipped item. To do this, UPS relies on a company-wide information system. Shipped items are the...
-
Draw resonance structures for this anion. Remember, sulfur can have 10 or even 12 electrons in its valence shell. :0: 24 CH-S2+0 :0:
-
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the following structure are in accord with predictions based on these resonance...
-
For the data presented in BEH.6, show the financial statement presentation of the securities and related accounts. Assume the securities are noncurrent. Data From BEH.6 In its first year of...
-
What is the rate of return for a $\$ 14,000,20$-year investment that provides a payout of $\$ 1,000$ a year?
-
A homeowner found a preferred savings account book in the wall of a home he was remodelling. He went to the bank and discovered that the account was 25 years old and had $\$ 425,801$ in the bank! The...
-
A lens has a radius of curvature of magnitude \(R\) on its left surface and a radius of curvature of magnitude \(2 R\) on its right surface. Parallel rays entering the lens from the left are focused...
-
What is the payback period for the project described below? Machine investment $24,999 MARR 18% Annual benefit $5,000 Annual maintenance Life Salvage value $2,500 7 years $1,000
-
Explain, through an example, how methods can be derived from messages.
-
\(\sigma_{1}
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
Determine whether the series is convergent or divergent. 8 n=1 cos n n cos 1 1 + cos 2 2 + cos 3 3 +
-
Draw the five cycloalkanes with the formula C5H10.
-
Draw two constitutional isomers of cis-1, 2-dibromo-cyclopentane.
-
Draw a stereoisomer of trans-1, 3-climethylcyclobutane.
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


