Question: Draw resonance structures for this anion. Remember, sulfur can have 10 or even 12 electrons in its valence shell. :0: 24 CH-S2+0 :0:
Draw resonance structures for this anion. Remember, sulfur can have 10 or even 12 electrons in its valence shell.
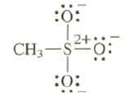
:0: 24 CH-S2+0 :0:
Step by Step Solution
3.56 Rating (156 Votes )
There are 3 Steps involved in it
0 21 CH3SO 0 ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-C (127).docx
120 KBs Word File


