Photolysis of the cis-Cyclobutene isomer in Problem 30.25 yields cLc-cyclododecaen-7-yne, hut photolysis of the trans isomer yields
Question:
Photolysis of the cis-Cyclobutene isomer in Problem 30.25 yields cLc-cyclododecaen-7-yne, hut photolysis of the trans isomer yields trans-cyclododecaen-7-yne. Explain these results, and identify the type and stereochemistry of the pericyclic reaction.
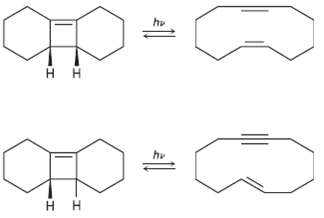
Transcribed Image Text:
hv нн hи нн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 22% (9 reviews)
H H hv hv go H H Both ...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why the following reaction yields the Hofmann product exclusively (no Zaitsev product at all) even though the base is not sterically hindered: Br NaOEt ELOH
-
Reaction of benzanilide with chlorine in acetic acid yields a mixture of two monochloro derivatives formed by electrophilic aromatic substitution. Suggest reasonable structures for these two isomers....
-
The relative rate of reaction of the cis alkene (E) is given in Table 24.2. What would you expect the relative rate of reaction of the trans isomer to be?
-
(a) Decide whether each of the following expressions are true or not. Answer yes or no. In any case where it is not true, provide the actual O-complexity. (i) log n = O(n!) (ii) 10! = O(log n) (iii)...
-
Whose interests do you believe antitrust regulators represent? What trade-offs do antitrust regulators face in making decisions that impact the groups whose interests they represent? Be specific.
-
The mean free path of a molecule in a gas is 300 nm. What will the mean free path be if the gas temperature is doubled at (a) Constant volume (b) Constant pressure?
-
Use R to create a randomization scheme to randomize 222 subjects to three treatments such that there are an equal number of subjects assigned to each treatment. You may use the functions developed in...
-
Suppose that Bigco is currently trading for $100 per share. We know that in one year Bigco stock will sell for either $150 per share (good day) or for $75 per share (bad day). No other prices are...
-
P 3 4u k2 1 12 x2 b A simple representation of a vehicle suspension system is shown in Figure above. Assume that the forces produced by the springs and dampers act at the end points P and Q and that...
-
SecuriCorp operates a fleet of armored cars that make scheduled pickups and deliveries in the Los Angeles area. The company is implementing an activity-based costing system that has four activity...
-
Ring-opening of the trans-Cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of the cis-Cyclobutene isomer. Explain the temperature effect, and identify the...
-
Propose a pericyclic mechanism to account for the followingtransformation: Heat HO. H.
-
Stanton Corp. began operations on January 1, 2016. The statement of cash flows for the first year reported dividends paid of $160,000. The balance sheet at the end of the first year reported $40,000...
-
Consider the following benefit and cost data of a project Year Year 0 Year Year Year Year 4 Year 5 Year 6 1 2 3 Benefits ($) 0 Costs ($) 5,000 8,000 9,000 12,000 10,000 12,000 16,000 2,000 4,000...
-
Given that the following Zero rate with semiannual compounding: Maturity (years) Zero rate(semi-annual) 0.5 1.0 5 5.25 1.5 2.0 5.5 6 (a) What are the rates with continuous compounding? (8 points) (b)...
-
Describe each of the research methods questionary, interviews, observation, and documents and images and when it might be best to employ it, from Martyn Denscombe's book.
-
The US Department of Homeland Security (DHS) Doctrine defines internal risks as follows Financial stewardship Malicious activity in cyberspace Personnel reliability Systems reliability
-
The entries to record cost and sale of a finished good on account is Group of answer choices debit Cost of Goods Sold, credit Finished Goods debit Cost of Goods Sold, credit Finished Goods, debit...
-
In which trading centers does 75 percent of all foreign exchange trading take place?
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Suppose that you are a trader at a large trading firm. You have just received a sell order from a mutual fund for 1,000,000 shares of stock that has an average daily volume of 250,000 shares. What...
-
Oleic acid and stearic acid are naturally occurring compounds, which can be isolated from various fats and oils. In the laboratory, each can be prepared by hydrogenation of a compound known as...
-
Sodium-ammonia reduction of stearolic acid (see Problem 9.10) yields a compound known as elaidic acid. What is the structure of elaidic acid?
-
Suggest efficient syntheses of (E)- and (Z)-2-heptene from propyne and any necessary organic or inorganic reagents.
-
On August 1, 2015, Lifters, a gym, sells 5 annual memberships for $1,500 each. What would the entry look like to record the receipt of cash as deferred revenue?
-
Lyme Incorporated issues 3,000 shares of no-par-value common stock for $20 per share. What is the correct journal entry for this transaction?
-
Unsubstantiated Expenses Your client, Randy Ross, owns a business that sells hinges, brackets, and fuses used in the manufacturing of machinery. Randy spends a large amount of time traveling to...

Study smarter with the SolutionInn App


