Ring-opening of the trans-Cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of
Question:
Ring-opening of the trans-Cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of the cis-Cyclobutene isomer. Explain the temperature effect, and identify the stereochemistry of each reaction as either conrotatory ordisrotatory.
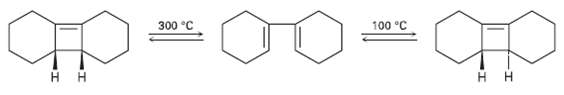
Transcribed Image Text:
100 °C 300 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
HH HH conrotatory conrotatory Ring opening of the tra...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A gaseous reaction takes place at constant volume and constant pressure in a cylinder shown here. Which of the following equations best describes the reaction? The initial temperature (T1) is twice...
-
A chemical reaction takes place in a container of cross-sectional area 50.0 cm/. As a result of the reaction, a piston is pushed out through 15 cm against an external pressure of 121 kPa. Calculate...
-
Explain why the reaction shown in eq. 7.19 occurs much more easily than the reaction (That is, why is it necessary to protonate the alcohol before ionization can occur?) (CH) C-OH(CHCHO
-
At the beginning of the year, Plummer's Sports Center bought three used fitness machines from Advantage, Inc. The machines immediately were overhauled, installed, and started operating. The machines...
-
Speculate as to why the share prices of American and US Airways increased sharply on the day that the agreement with the Justice Department had been reached? Why did the share prices of other major...
-
The density of air at STP is about 1/1000 the density of water. How does the average distance between air molecules compare to the average distance between water molecules? Explain.
-
Explain when it is appropriate to use a randomized block design.
-
1. Which promotional mix elements does Snickers use? 2. How does this Snickers campaign demonstrate the characteristics of integration? 3. What grade would you give Snickers on integration...
-
Hazelnut Corporation manufactures lawn ornaments. It currently has two product lines, the basic and the luxury. Hazelnut has a total of $169,122 in overhead. The company has identified the following...
-
A loan of $7,419 was repaid at the end of 14 months. What amount of repayment check (principal and interest) was written, if a 4.7% annual rate of interest was charged?
-
Predict the product of the following pericyclic reaction. Is this [5, 5] shift a suprafacial or an antarafacialprocess? [5,5] CH eat
-
Photolysis of the cis-Cyclobutene isomer in Problem 30.25 yields cLc-cyclododecaen-7-yne, hut photolysis of the trans isomer yields trans-cyclododecaen-7-yne. Explain these results, and identify the...
-
For a new product, sales volume in the first year is estimated to be 80,000 units and is projected to grow at a rate of 4% per year. The selling price is $ 12 and will increase by $ 0.50 each year....
-
what ways can governmental policies and cultural initiatives support the recognition, documentation, and safeguarding of folk art traditions, particularly those facing threats from urbanization,...
-
How do folk art festivals and exhibitions serve as platforms for fostering intercultural dialogue, promoting cross-cultural understanding, and revitalizing interest in traditional artistic practices?
-
How do regional variations in folk art reflect the diverse histories, beliefs, and lifestyles of different communities, and what role does cultural exchange play in shaping these artistic expressions?
-
What cultural significance do indigenous folk art traditions hold within their respective communities, and how have they evolved over time amidst modernization and globalization?
-
How did the reception and interpretation of classical art vary across different cultural and geographical contexts, and what strategies did artists and scholars employ to reconcile the tension...
-
What is the purpose of a currency option?
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Long-term U.S. Treasury bonds are considered to be very high-quality investments with virtually zero default risk. Is it reasonable to assume that the risk of settling U.S. Treasury bonds is also of...
-
Suggest a structure for the product of nucleophilic substitution obtained on solvolysis of tert-butyl bromide in methanol, and outline a reasonable mechanism for its formation.
-
Identify the compound in each of the following pairs that reacts at the faster rate in an SN1 reaction: (a) Cyclopentyl iodide or 1-methylcyclopentyl iodide (b) Cyclopentyl bromide or...
-
Use curved arrows to show how calcium carbide reacts with water to give acetylene.
-
Many of us pay close attention to and interpret nonverbal messages more easily when communicating face to face, but in todays world, much of our communication is done virtually. Why is your ability...
-
A taxpayer who is an employee generally has what kind of taxes withheld from his wages? Explain
-
How do population genetics and molecular ecology contribute to our understanding of adaptive evolution, particularly in the context of rapidly changing environments and human-mediated disturbances?

Study smarter with the SolutionInn App


