Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the
Question:
Predict the geometry of the following compounds at the indicated atoms:
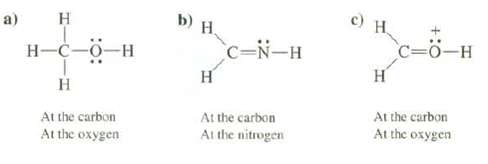
Transcribed Image Text:
a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
a Tetrahedral at C bent at ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the geometry of the following molecules and ion using the VSEPR model: (a) CH3I, (b) ClF3, (c) H2S, (d) SO3, (e) SO42-?
-
Predict the geometry of the following ions, using the electron-pair repulsion model. a. ClO3 b. PO43 c. SCN d. H3O+
-
Predict the geometry of the following molecules using the VSEPR method: (a) HgBr2, (b) N2O (arrangement of atoms is NNO), (c) SCN- (arrangement of atoms is SCN)?
-
Which of the following options are available for creating a policy in Qualys Policy Compliance? (Choose three) A, Create from Host B, Create from Scratch C, Import from Library D, Import from CSV File
-
Monitor Ltd uses 1680 tonnes of a chemical each year in its manufacturing processes. Monthly demand for the chemical fluctuates between 110 and 160 tonnes. The lead time for each order is one month,...
-
What is utility? How does production create utility?
-
The Haaland formula for the friction factor is \[ f=\frac{0.3086}{\left\{\log \left[6.9 / \operatorname{Re}+(\varepsilon / 3.7 D)^{1.11} ight] ight\}^{2}} \] Compare this equation for \(f\) for...
-
Granville Community College is considering outsourcing the maintenance of its buildings and other facilities to an outside rm for $300,000 per year. The 2013 budget is as follows: Granville...
-
Write a Javascript program to display the list of Student names whose name starts with 'A' For example, you have an array of students names Khadija Ali ahmed List of name starts with alphabet 'A' or...
-
Locate all the instantaneous centres for the crossed four bar mechanism as shown in Fig. 6.28. The dimensions of various links are : CD = 65 mm; CA = 60 mm ; DB = 80 mm ; and AB = 55 mm. Find the...
-
Predict the geometry at the carbon of these compounds: a) H-C=N: b) 0: H-C-0-H c) H T CI-C-H T CI
-
Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H H H-C-C=N H
-
Using estimates of rainfall, evaporation, and water consumption, the town engineer developed the following model of the water volume in the reservoir as a function of time where V is the water volume...
-
As you think about the new health care reform bill, what type of lawmaking action instituted it? In other words, was it developed from the common law, executive order, or legislative process? Was...
-
What motivational strategy would you apply with an employee who you think is capable of doing the work but is underperforming?
-
Why have conventional indemnity policies been supplanted by other forms on health insurance coverage?
-
Which myth of motivation is the most important? Are there other myths you can identify?
-
What are the pros and cons of the different types of health insurance benefit packages that someone might purchase?
-
Who discovered that energy and mass are two different forms of the same thing?
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
Find f g h if f(x) = x/(x + 1), g(x) = x 10 , and h(x) = x + 3.
-
Propose structures for molecules that meet the following descriptions: (a) Contains two sp2-hybridized carbons and two sp3-hybrized carbons (b) Contains only four carbons, all of which are...
-
Why cant molecules with the following formulas exist? (a) CH5 (b) C2H6N (c) C3H5Br2
-
Draw a three-dimensional representation of the oxygen-bearing carbons atom in ethanol, CH3CH2OH, using the standard convention of solid, wedged, and dashed lines.
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...
-
An 84 year old female arrives to the ER today with her son with a complaint of new onset of confusion. What are your differential diagnoses for a patient with new altered mental status? Go through...

Study smarter with the SolutionInn App


