Predict the products of the following Diels?Alder reactions; (b) (a)
Question:
Predict the products of the following Diels?Alder reactions;
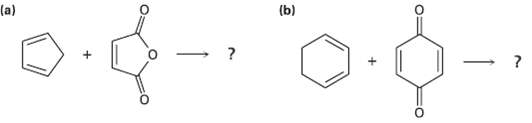
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a H O 11 00 b If two equiva...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) Excess NH3 + Ph - CH2CH2CH2Br (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) (I) NaN3 (2) LiAIH (3) H30 1-bromopentane CH3...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Cliff Arthur has equally attractive job offers in Miami and Los Angeles. The rent ratios in the cities are 8 and 20, respectively. Cliff would really like to buy rather than rent a home after he...
-
What can a company gain from union-management cooperation? What can workers gain?
-
Design an RL lowpass filter that uses a 40-mH coil and has a cutoff frequency of 5 kHz.
-
Design a process for the separation of \(1,000 \mathrm{kgmol} / \mathrm{hr}\) of a mixture composed of \(50 \mathrm{~mol} \% \mathrm{~A}\) and \(50 \mathrm{~mol} \% \mathrm{~B}\) (water) to product...
-
On May 12, 2014, Chewco Co. purchased 2,000 shares of Jedi Inc. for $112 per share, including the brokerage commission. The Jedi investment was classified as an available-for-sale security. On...
-
ASL Ltd's most recent earnings per share was $1.00 and it paid a dividend of $0.60 per share. You expect ASL to earn $1.10 and continue its payout ratio. Assume that you expect to sell the shares for...
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, objectivity, or credibility)....
-
Cody is in college. How do you think his Statement of Financial Position will change after he graduates from college? Identify at least two specific changes.
-
Using your own company accomplish the following: I. Company Name and Its Background (give brief description: the company background, its firmographic profile, business operations, summary only) II....
-
49. Given the following air sample data, calculate the gross activity airborne concentration in Ci/em": frisker bkg = 200 cpm air sample volume = 60 ft 1 Ci 2.22 x 10 dpm gross count rate on filter =...
-
Chang Company has a December 31 year end. In year 7 it bought a piece of equipment at the start of the year for $572,500 and employed straight line depreciation over 5 years with an estimated...
-
Let x(n) = {0, 1, 2, 3}. 1. Compute its discrete-time Fourier transform X(e). 2 2. Sample X(e) at ka = k, k = 0,1,2,3. 4 Show that it is equal to X (k).
-
Solve the IVP. Do not use Laplace transformation. \(\ddot{y}-y=t^{2}-\cos t, \quad y(0)=\frac{1}{2}, \dot{y}(0)=0\)
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
Solution A is a 1.0 m solution with a nonionic solute and water as the solvent. Solution B is a 1.0 m solution with the same nonionic solute and ethanol as the solvent. Which solution has the greater...
-
The antioxidants BHA and BHT are commonly used as food preservatives. Show how BHA and BHT can be made from phenol and hydroquinone. C(CH33 (CHC CICH CH OCH3 BHA BHT
-
Propose a mechanism for the bromination of ethoxybenzene to give o- and p-bromoethoxybenzene.
-
Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears...
-
1. Mention five key organizational characteristics that the Cll research team identified for organizations with effective quality management systems. provide brief discussion of each and how it...
-
When a metal was exposed to photons at a frequency of 1.46 1015 s1, electrons were emitted with a maximum kinetic energy 3.60 10-19 J. G ? Calculate the work function, , of this metal. J/photon What...
-
No. 101 Debit Cash $ 2,400 Question 2 (10 pts) On November 1, 2024, the account balances of Schilling Equipment Repair were as follows. Accumulated Depreciation-Equipment A Clipboard Font Alignment...

Study smarter with the SolutionInn App


