Propose a synthesis of the drug sulfathiazole from benzene and any necessaryamine. Sulfathiazole H2N
Question:
Propose a synthesis of the drug sulfathiazole from benzene and any necessaryamine.
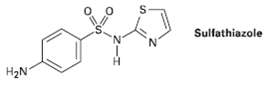
Transcribed Image Text:
Sulfathiazole H2N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
This reaction sequence is similar to the sequence used to synthesize sulfanilamide Key s...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a synthesis of 4, 4-dimethyl-2-pentyne (the compound in Problem 14.19) from an alkyl halide and an alkyne.
-
Propose a synthesis of the anti-inflammatory drug Fenclorac fromphenyl-cyclohexane. CI Fenclorac CHCO2H
-
Propose a synthesis of (R)-CH3CHN3CH2CH3, starting from (R)-2-chlorobutane.
-
Match the following ratios with the appropriate formula. Ratio or Rate Formula a. Income from operations Interest expense Acid-test Total liabilities Stockholders' equity Current b. Net income-...
-
Do you agree with how Adam Silver handled the situation? Do leaders really set the tone of an organization's values?
-
A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass fractions of 0.5 for both and phases. If the composition of the phase is 90 wt% B-10 wt%...
-
A compound is produced for a coating process. It is added to an otherwise fixed recipe and the coating process is completed. Adhesion is then measured. The following data concern the amount of...
-
Given the following information regarding a project concerning an initial public offering (IPO), (a) Draw the network. (b) What is the critical path? (c) When will the offering be available...
-
1. Identify a specific business that uses Artificial Intelligence and Robotics. 2. Why did you select this organization for your research project? 3. Identify three major issues for the use of...
-
1. Which process should VBB choose to produce?? 2. How much would VBP be willing to pay for the testing that is currently offered, for each batch?? 3. Would we be considered a perfect test, at twice...
-
What products would you expect from Hofmann elimination of the following amines? If more than one product is formed, indicate which ismajor. NH2 (b) NH2 (a) CH3CH2CH2CHCH2CH2CH2CH3 NHCH2CH3 (d) NH2...
-
Propose syntheses of the following compounds from benzene: (a) N, N-Dimethylaniline (b) p-Chloroaniline (c) m-Chloroaniline (d) 2, 4-Dimethylaniline
-
Which of the following C corporations will be allowed to use the cash method of accounting for 2021? Explain your answers. a. Jade Corporation, which had gross receipts of $26,300,000 in 2018,...
-
Fox anticipates that there is a good chance of his bond being called after two years and that the issuer will pay a penalty of $50 per $1,000 of the face amount.What is its yield to call?
-
Twin Oaks Brewery (TOB) needs to borrow $20 million immediately. It can borrow for three years at a fixed rate of 7.5% or at a floating rate of LIBOR + 40 basis points. Plain vanilla...
-
2 For the function f(x) = x + 5x-14 solve the following. f(x) 0
-
You form a short straddle by selling a call with a premium of C = $9, and selling a put with a premium of P = $8. Both options have an exercise price of X = $47, both mature in 8 months, and both...
-
You started a company that manufactures belts and sells them for $200 each. If you need to sell 2000 belts per month to break even and fixed costs per month are $30000, what is the variable cost per...
-
BioElectroMechanical Systems (BEMS) is a start-up company with high potential and little available cash. They obtain $500,000 for necessary technology from a venture capitalist who charges them 24...
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
At 70 K, CCl 4 decomposes to carbon and chlorine. The K p for the decomposition is 0.76. Find the starting pressure of CCl 4 at this temperature that will produce a total pressure of 1.0 atm at...
-
Divide the following compounds into groups that might be expected to exhibit similar chemical behavior: a. C4H10 b. CH3OCH3 c. C3H7OH d. C8H18 e. HOCH2CH2CH2OH f. CH3NH2 g. CH3CH2CH3 h. CH3OH i....
-
Write an equation similar to eq. 1.2 for the formation of a fluorine molecule from two fluorine atoms.
-
Using Table 1.6, write a structural formula for each of the following: a. An alcohol, C3H8O b. An ether, C4H10O c. An aldehyde, C3H6O d. A ketone, C3H6O e. A carboxylic acid, C3H6O2 f. An ester,...
-
Zach Ltd. produced the following data for the year 2015 - o Interest Income $4,650,000 o Capital Gain realized $50,000 o Dividend Income from company's 15% ownership in XYZ Ltd. $65,000 o Corporate...
-
A project requires an increase in net working capital of $150,000 at time 0 that will be recovered at the end of its 10 year life. If the opportunity cost of capital is 8%, what is the effect of NWC...
-
A firm is 40% financed by debt with a yield-to-maturity of 8.5%. The equity has a beta of 1.3, the market risk premium is 8.4% and the risk-free rate is 3.8%. What is the firm's WACC if the tax rate...

Study smarter with the SolutionInn App


