Propose a mechanism for the followingreaction: HO- CO2CH3 (CH3CH2)3N Heat BRCH2 CO2CH3
Question:
Propose a mechanism for the followingreaction:
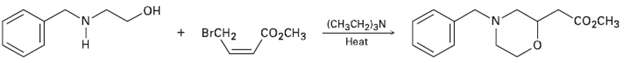
Transcribed Image Text:
HO- CO2CH3 (CH3CH2)3N Heat BRCH2 CO2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
NH HC proton transfer N Br COCH...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Propose a mechanism for the base-promoted hydrolysis of g-butyrolactone:
-
Suppose a single 802.11n client is connected to an 802.11n access point and there is no other client or access point in the neighborhood. The client senses the medium, then sends a 2000-Byte frame,...
-
What information or questions can you use when writing a follow-up message after submitting a résumé?
-
Walker owned several pizza parlors that operated under the name of El Fredo Pizza, Inc. He planned to open a new parlor and to purchase a new oven. When a friend suggested that Walker purchase an...
-
A sociologist surveys a sample of college students to determine whether there are differences in the attitudes and behaviors of male and female students. The survey contains 20 questions. For one...
-
The following data were taken from the general ledger and other data of McDonough Manufacturing on July 31: Finished goods, July 1................................................... $ 85,000 Cost of...
-
Rate on Taxable Income 10% 12 22 24 33 35 37 Single Taxpayers Up to $9,525 $9,525-$38,700 Married Taxpayers Filing Jointly Up to $19,050 $19,050-$77,400 $38,700-$82,500 $77,400-$165,000 Head of...
-
Throughout the course, you will be working on a Wealth Management Analyst Project For this project, imagine you are a new hire at a wealth management firm and tasked with determining the location of...
-
The following transformation involves a conjugate nucleophilic addition reaction (Section 19.13) followed by an intra molecular nucleophilic acyl substitution reaction (Section 21.2). Show...
-
One step in the biosynthesis of morphine is the reaction of dopamine with p-hydroxyphenylacetaldehyde to give S)-norcoclaurine. Assuming that the reaction is acid-catalyzed, propose amechanism. . NH ...
-
Define the following and explain why they are important: burn rate, free cash flow, fume date, time to clear, and financial management myopia.
-
A firm seeking rapid growth should pursue __________. A. internal growth B. external growth C. divestment of poor performing businesses D. a restructuring strategy
-
Goals are specific and often quantified versions of objectives. True or False
-
If a firm is able to consistently earn above-average profits, it is effectively balancing the goals of its stakeholders. True or False
-
Which of the following is not a characteristic of strategic decisions? A. They are long term in nature. B. They involve choices. C. They do not involve trade-offs. D. All of the above are...
-
Explain how changes in interest rates affect the automobile, home construction, and auto repair industries.
-
Things looked good for Mylan NV, a leading generic pharmaceutical drug maker, midday on November 13, 2015, as the early results of the firms campaign to acquire Perrigo appeared promising. The votes...
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
Find the pH of a 0.0100 M sulfuric acid (H 2 SO 4 ) solution.
-
Write out the steps in the mechanism for the sulfonation of benzene.
-
Which product would you expect if propene were used in place of ethene in eq. 4.11 (or eqs. 4.20 and 4.21): propylbenzene or isopropylbenzene? Explain. Eq. 4.11 CH2CH3 H2SO alkylation (4.11)
-
Draw the important resonance contributors for the benzenonium intermediate in the bromination of aniline, and explain why ortho, para substitution predominates. NH2 aniline
-
Compare and contrast digital evidence and non-digital evidence. Provide three examples of how they are similar and three examples of how they are different. You must use the provided course materials...
-
In May of 2015, Mr. Miradi made an oral agreement with Mr. Miondoko for the sale of a piece of land situate in Nyeri County Land title No. Gakawa/Kahurura Block III/Mwichwiri/823 for a price of KShs....
-
and QB- Two identical point charges (+2.27 10-9 C) are fixed in place, separated by 0.460 m (see the figure). Find (a) the electric field and (b) the electric potential at the midpoint of the line...

Study smarter with the SolutionInn App


