Question: Repeat problem for the network shown infigure. R1 vslt) (1)a R23 10
Repeat problem for the network shown infigure.
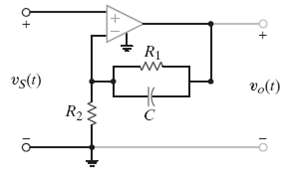
R1 vslt) (1)a R23 10
Step by Step Solution
3.44 Rating (183 Votes )
There are 3 Steps involved in it
Z Z 2 Z R 7 Gv vo R It JWR C ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
27-E-E-C-A (1874).docx
120 KBs Word File


