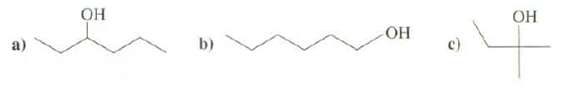
Show preparation of these alcohols fromalkenes. b) c)
Question:
Show preparation of these alcohols fromalkenes.
Transcribed Image Text:
ОН ОН он b) c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
a b c 1 HgOCC...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show what alcohols and carbonyl compounds give the following derivatives. (a) (b) (c) (d) (e) (f) CH CH,O OCH,CH CH O-CH CH3 CH3 CH-C H O-CH CH a,0 OX
-
Show how you would synthesize the following alcohols from appropriate alkenes. (a) (b) (c) (d) ,
-
Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde. (a) (b) (c) CH,OH CH,OH
-
Consider the following population regression model: y = Bo + Bx + Bx2 + 3x3 + u Suppose you want to test whether 0.532 = 83. The hypotheses are: Ho : 0.582 = 33 H : 0.532 #33 The correct expression...
-
Management accountants need a wide variety of skills for their roles in organizations. These skills can be classified as either technical or nontechnical. Required: For each of the following skills,...
-
27) Alfa school started imparting education on 1.4.2010, with the sole objective of providing education to children of weaker society either free of cost or at a very nominal fee depending upon the...
-
When is DT&E performed during the System Development Phase?
-
The comparative balance sheet of Middaugh Restaurant Supplies Inc. for December 31, 2013 and 2012, is as follows: The income statement for the year ended December 31, 2012, is as follows: The...
-
What is the relationship between organizational investment in employee development programs and the resultant levels of organizational commitment ?
-
Mirza Manufacturing makes four electronic products, each of which comprises three main materials: magnet, wiring, and casing. The products are shipped to three distribution centers in North America,...
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Find the 15th term of the arithmetic sequence 2, 5, 8, c.
-
Sandy Grossman (potential Nobel laureate) gets a job at Wharton Business School (Upenn) for an undisclosed salary. At his inaugural the School's provost says "I am delighted to introduce Sandy to you...
-
What is capital rationing? What types are there? What problems does capital rationing create for discounted cash flow analysis?
-
Suppose Fraser Banks rate-sensitive assets are $10m securities, and its rate-sensitive liabilities are $25m chequable deposits. What will happen to its profit if the interest rate changes from 4% to...
-
What are the basic components of credit policy? What are the basic components of the terms of sale if a firm chooses to sell on credit?
-
The organization that Mike works in finds that one of their domains is directing traffic to a competitors website. When Mike checks, the domain information has been changed, including the contact...
-
A \(5.3-\mathrm{kg}\) ball is dropped from a height of \(2.0 \mathrm{~m}\) into a vat of water. The impact produces \(0.80 \mathrm{~J}\) of sound energy, the ball gains \(4.50 \mathrm{~J}\) of...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Calculate the pH after 0.10 mole of NaOH is added to 1.00 L of the solution in Exercise 31, and calculate the pH after 0.20 mole of HCl is added to 1.00 L of the solution in Exercise 31. Data in...
-
Chlorophyll, heme, vitamin B12, and a host of other substances are bio-synthesized from porphohilinogen (PBG), which is itself formed from condensation of two molecules of 5-aminolevulinate. The two...
-
Choline, a component of the phospholipids in cell membranes, can be prepared by SN2 reaction of trimethyl amine with ethylene oxide. Show the structure of choline, and propose a mechanism for...
-
Cyclopentamine is an amphetamine-like central nervous system stimulant. Propose a synthesis of Cyclopentamine from materials of five carbons orless. CH3 -CH2CHNHCH3 Cyclopentamine
-
Marigold, Inc. reports all its sales on credit, and pays operating costs in the month incurred. Amounts for 2022 are: March April May June July Budgeted sales $299500 $290000 $319700 $279100 $209800...
-
4. Superman is very strong. In trying to impress Lois Lane, he stands on top of a 12-storey building (-50 meters tall), lowers a very tall and wide straw into a pool of Kool-Aid and tries to drink....
-
How is the rise of the gig economy reshaping traditional organizational structures, and what are the implications for workforce management and employee engagement ?

Study smarter with the SolutionInn App


