Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2)
Question:
Show the products of thesereactions:
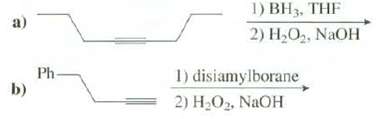
Transcribed Image Text:
1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
a...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
For the curve defined by F(t) = (etcos(t), e* sin(t)) find the unit tangent vector, unit normal vector, normal acceleration, and tangential acceleration at 5A t 6 T N (5r) = 6 5T 6 aN ||
-
Describe the impacts of ADS systems.
-
For a home repair job you must turn the handle of a screwdriver 27 times. (a) If you apply an average force of 13 N tangentially to the 3.0-cm-diameter handle, how much work have you done? (b) If you...
-
Traders in major financial institutions use the Black-Scholes formula in a backward fashion to infer other traders' estimates of \(\sigma\) from option prices. In fact, traders frequently quote...
-
The following is taken from the Colaw Company balance sheet. Interest is payable annually on January 1. The bonds are callable on any annual interest date. Colaw uses straight-line amortization for...
-
1. How would you describe the basic components of WACC to a group of decision makers in a company? 2. On the most basic level, if a firm's WACC is 12 percent, what does this mean? 3. In calculating...
-
Sima Donuts supplies its retail outlets with the ingredients for making fresh donuts. A central warehouse from which trucks are dispatched is located in Atlanta. Trucks can leave the Atlanta...
-
Show preparation of these alcohols fromalkenes. b) c)
-
Show syntheses of these compounds from1-bromobutane: b) TH. c)
-
Two particles travel along the space curves r(t) and u(t). A collision will occur at the point of intersection P when both particles are at P at the same time. Do the particles collide? Do their...
-
A force of ( 2 * 1 0 0 ) N makes an angle of 1 1 degrees with the direction of travel. Find the work done if the force moves 9 4 m . A machine rotates at ( 8 8 * 1 0 0 ) rpm and decelerates at 2...
-
Moderator in a nuclear reactor. When fast neutrons are produced in a nuclear reactor, they must be slowed before they can participate in a chain-reaction process. This is done by allowing them to...
-
In a rotating vertical cylinder (Rotor-ride) a rider finds herself pressed with her back to the rotating wall. Which is the correct free-body diagram for her? HX1 (d) (a) (b) (c) (e)
-
A merry-go-round has an angular velocity of 5.60 rad/s. A ribbon is tied at the end of one of the arms and it has speed of 3.40 m/s. At what distance is the ribbon from the center of the...
-
The imaginary line perpendicular to the incident ray onto a materials interface is known as a Tangent line Normal line Skewed line Bent line
-
Ask older friends or relatives about the cost of specific items (e.g., a gallon of gas, a cup of coffee, etc.) during their youth. Also inquire bout their average wages in the past. Compare the...
-
Is it a breach of fiduciary duty for a director of a real estate investment trust (REIT) negotiating a joint venture on behalf of the REIT with another director for the development of a portfolio of...
-
Calculate the pH of a solution that is 1.00 M HNO 2 and 1.00 M NaNO 2 .
-
Tetracaine is a substance used medicinally as a spinal anesthetic during lumbar punctures (spinal taps). (a) How would you prepare tetracaine from the corresponding aniline derivative, ArNH2? (b) How...
-
Atropine, C 17 H 23 NO 3 , is a poisonous alkaloid isolated from the leaves and roots of Atropa belladonna, the deadly nightshade. In small doses, atropine acts as a muscle relaxant; 0.5ng (nanogram,...
-
Tropidene (Problem 24.58) can be converted by a series of steps into tropili-dene (1, 3, S-cycloheptatriene). How would you accomplish this conversion?
-
World Tourism Day 2 0 1 7 , with its official celebration on September 2 7 in Qatar, was focused on sustainable tourism. Established by the United Nations World Tourism Organization ( UNWTO ) , World...
-
Tracy is single and had adjusted gross income of $35,800 in 2018. Tracy also has the following items: How much may Tracy claim as itemized deductions?
-
Calculate the geometric mean return of an investment with five year returns of 10%, (15%), 12%, 8% and (5%).

Study smarter with the SolutionInn App


