Show syntheses of these compounds from1-bromobutane: b) TH. c)
Question:
Show syntheses of these compounds from1-bromobutane:
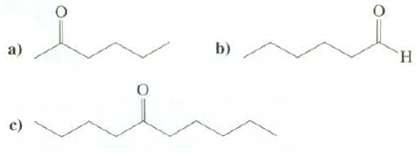
Transcribed Image Text:
b) TH. c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a b h HO HSO4 HgSO4 ...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indication starting materials. Your syntheses may produce both enantiomers of any target that ischiral. a) CH.CH,CH,CH,CH,OCH, from CH;CH,CH,CH=CH2 CH3 b)...
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Show syntheses of these compounds from the indicated startingmaterials. a) CH,CH,CH,CH,N(CH3), from compounds with none of the CN bonds of the final product. CH3 b) CH,CH,OCHCH,CH; from alkyl halides...
-
Consider the following molecule: Tin II Chloride(SnCl2) Are the bonds polar or non-polar? Is the molecule polar or non-polar?
-
Define Web 2.0.
-
If an EDM instrument has a purported accuracy capability of (3 mm + 3 ppm), what error can be expected in a measured distance of? (a) 30 m (b) 1586.49 ft (c) 975.468 m (Assume that the instrument...
-
Charlie and Larry both face the same budget line for consumption and leisure. At every possible consumption-leisure bundle on the budget line, Charlie always requires marginally more leisure than...
-
Penny Davis runs the Shear Beauty Salon near a college campus. Several months ago, Penny used some unused space at the back of the salon and bought two used tanning beds. She hired a receptionist and...
-
137- We want to use K-Means with K=2 for the following data. Where should we initially place the cluster centers? Subject A B 1 1.0 1.0 2 1.5 2.0 34 3 3.0 4.0 4 5.0 7.0 5 3.5 5.0 19 6 4.5 5.0 7 3.5...
-
IFRS defines intangible assets as lacking physical substance, non-monetary in nature, and identifiable. For the following items, identify the reason or reasons why the item is not considered an...
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Show the products of thesereactions: CH CH;CH C3D CH CH3 .. -- NaOH CH3
-
When the items needed for the additional information required by the direct method are so easy to identify from the Cash Book, why do you think most companies prefer to use the indirect method?
-
A company earns 15 cents (after interest) per share and pays a regular dividend of 25 cents per share. The other 5 cents is kept in the business to help reduce gearing and will earn 3% per annum. The...
-
Overall, does Tesla have a good strategy? Why or why not? How do you know? Explain.
-
Which of these molecular formulas are also empirical formulas? aspirin (C9H8O4) ethyl ether (C4H10O) benzene (C6H6) ethyl acetate (C4H8O2) glucose (C6H12O6) citric acid (C6H8O7)
-
Corrected Journal Entries, Corrected Inventory, Ledger Accounts, Income Statement, and Balance Sheet. The Ledger Accounts, Income Statement, and Balance Sheet tabs should be based on the Corrected...
-
A charge +q is located at 3.000 cm, a second charge -2q is located at 8.000 cm, and a third charge +q is located at 11.00 cm. The magnitude of q is 2.450 C. + +q 5.000 -29 What is the force F, on the...
-
Describe how the legislative branch of government is a source of law.
-
What are the 5 Cs of marketing channel structure?
-
Instead of the titration of a strong acid by a strong base considered in Question 5, consider the titration of a strong base by a strong acid. Compare and contrast a strong acidstrong base titration...
-
Propose a structure for the product with formula C9H17N that results when 2-(2-cyanoethyl) Cyclohexanone is reducedcatalytically. CH2CH2CN H/Pt C9H17N
-
Coniine, C8H17N, is the toxic principle of the poison hemlock drunk by Socrates. When subjected to Hofmann elimination, coniine yields 5-(N, N-dimethylamino)-1-octene. If coniine is a secondary...
-
How would von synthesize coniine (Problem 24.61) from acrylonitrile (H2C = CHCN) and ethyl 3-oxohexanoate (CH3CH2CH2COCH2CO2Et)?
-
S&J Catering is a small catering business operating in Western Sydney. Business partners, Jack and Simon, established the business a year ago. To start up the business, the partners contributed...
-
In 20X1, Lee was hurt in the course of his employment with Foster Farms. In 20X1, Lee was paid the following amounts from Foster Farms' worker's compensation insurance company: $30,000 for a physical...
-
Briefly describe tax administrative matters including: Tax collection and withholding mechanisms (PAYG withholding, PAYG instalments).
Effective C An Introduction To Professional C Programming 1st Edition - ISBN: 1718501048 - Free Book

Study smarter with the SolutionInn App


