Show the important resonance structures for these compounds. Use the curved arrow convention to show how the
Question:
Show the important resonance structures for these compounds. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure.
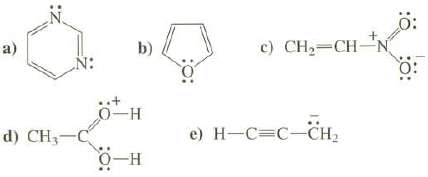
Transcribed Image Text:
N: d) CH₂-C b) 0-H O-H c) CH₂=CH-N e) H-C=C-CH₂ Ö: 0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a The unshared electron pairs on the nitrogens are not part of the conjugated pi system b One uns...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative...
-
Draw the important resonance structures for these species and discuss the contribution of each to the resonance hybrid. Explain whether the species has a large or a small amount of resonance...
-
You are developing an industrial building with a gross building area of 150,000 sf. The building efficiency ratio is 75%. The market gross rent is $25 psf. The vacancy rate is 5%; the cap rate is 5%;...
-
How might real-time reporting contribute to improving the competitive advantage of an organization?
-
Virtually all countries have universal government-run health-care systems. The United States is one notable exception. This is an issue in every election, with some politicians pushing for the United...
-
A statistics student has constructed a confidence interval for the mean height of daughters whose mothers are 66 inches tall, and a prediction interval for the height of a particular daughter whose...
-
NPV and AARR, goal-congruence issues. Nate Stately, a manager of the Plate Division for the Great Slate Manufacturing Company, has the opportunity to expand the division by investing in additional...
-
I believe that the construction of a DNA database containing the genetic information of every resident in the United States is a topic that demands careful consideration. Such a database holds the...
-
The frequency distribution shows the blood glucose levels (in milligrams per deciliter) for 50 patients at a medical facility. Construct a histogram, frequency polygon, and ogive for the data....
-
Draw the p orbital's that compose the conjugated part of these molecules: a) :-CH3 b) CH,=CHNH, c) H-C-C-CH=CH
-
In these examples the additional structure or structures are not important contributors to the resonance hybrid for the compound represented by the first structure, explain. a) 8-8 c) CH-C=N: b) :0...
-
Your boss is very puzzled by the finance courses in his MBA program. He has learned that cash flow is king, but notices that the capital budgeting problems spend a lot of time and effort dealing with...
-
Provide a brief summary of the following websites: Atwood, C. G. (2020). Succession planning basics (2nd edition.). ATD Press. Retrieved from...
-
Zhang Company leased equipment from Mann Industries. The lease agreement qualifies as a finance lease and requires annual lease payments of $47,847 over a six-year lease term (also the asset's useful...
-
When I am looking for an experimental Modulus of elasticity in a reinforced concrete Beam, where do I start my .1% offset on my load over the deformation graph? Is it .001 or .1% of the total...
-
1. Propel Corporation plans to make an investment, initially funded completely with debt. The investment requires an initial upfront cost of $49.8 million. The free cash flows of the investment and...
-
Essay Question 1 - Chapter 11 & Chapter 12 As discussed in the article by Lisa Bannon, the USC is being sued for promoting their online Masters in Social Work Classes as equivalent to USC's in-person...
-
How do angel syndicates work?
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
Indicate which tests should be used. n + 1 =1 33 + 4n? + 2
-
What are the relative energies of the three possible staggered conformations around the C2C3 bond in 2, 3-dimethylbutane? (See Problem 3.42)
-
Construct a qualitative potential-energy diagram for rotation about the CC bond of 1, 2-dibromoethane. Which conformation would you expect to be more stable? Label the anti and gauche conformations...
-
Which conformation of 1, 2-dibrornoethane (Problem 3.44) would you expect to have the larger dipole moment? The observed dipole moment of 1, 2-dibromoethane is = 1.0 D. What does this tell you about...
-
Investing all your wealth in the portfolio with the highest Sharpe Ratio is the strategy that will give you the best chance of meeting your goals. I. Yes II. No Why?
-
Bridgeport, Inc. produces stereo speakers. The selling price per pair of speakers is $1,000. The variable cost of production is $380 and the fixed cost per month is $45,260. For November, the company...
-
Donald, who has wages of $21,500, self-employment income of $31,725, discrimination lawsuit award of $22,500, and interest income of $650, has the following expenses for 2023: Alimony paid of $18,750...

Study smarter with the SolutionInn App


