Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4
Question:
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4
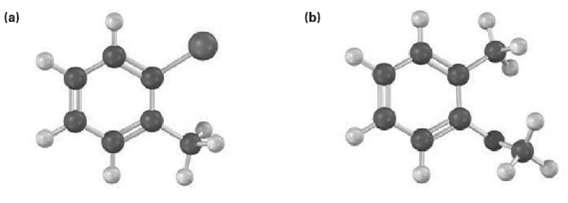
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a Br CH3 CH3 1 CH3CHCl AICI 3 OCH3 ii HNO3 HSO4 i CH3CHCl AICI 3 ii HNO3 HS...View the full answer

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall. H (a) . (b) CH3D2H
-
Show the major product for the following transformation: COOEt H3O" C10H100 heat (A) (B) (D) (C)
-
The structures of some common crystalline substances follow. Show that the net composition of each unit cell corresponds to the correct formula of each substance. CI Na Oa cs CI Ti
-
Suppose x = e y . Compute the value of y, given the following values of x: (a) x = 2 (b) x = 3 (c) x ~ 1.5 (d) x 3 (e) x = .5 (f) x = .002 (g) x = 10 (h) x = 1
-
In March 2016, Helen Carlon acquired used equipment for her business at a cost of $300,000. The equipment is five-year property for depreciation purposes. a. If Helen depreciates the equipment using...
-
Wildcat activity. Wildcats are wells drilled to find and produce oil and/or gas in an improved area or to find a new reservoir in a field previously found to be productive of oil or gas or to extend...
-
For each of the following, calculate the \(t\)-test for independent means. a. X 13.49,X 23.14, sX-1-X-2 = .31 b. X 113.27, X 2 = 16.45, sX 1-X 2 = 1.52 c. X 1.76, X 2.91, sX 1-X 2 = .09 d. X 11.52, X...
-
Selected accounts from Keystone Furnitures adjusted trial balance as of August 31, 2014, the end of the fiscal year, follow. Required 1. Prepare a multistep income statement for Keystone. Store...
-
May 1 G. Gram invested $43,000 cash in the company in exchange for its common stock. May 1 The company rented a furnished office and paid $2,200 cash for May's rent. May 3 The company purchased...
-
Are you optimistic about our ability to develop a global ethical consensus across our national and religious differences? Why or why not?
-
At what position would you expect electrophilic substitution to occur in each of the followingsubstances? (a) (b) (c) NH2 Br NO2 CI. Br
-
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism. FC. F3C. "CH-CH NO2 "CH-CH NO2 yfluorfen
-
Metro Technology began the year with inventory of $299,000 and purchased $1,820,000 of goods during the year. Sales for the year are $3,887,500, and Metro Technology's gross profit percentage is 60%...
-
An important initial step in establishing financial responsibility is to verify insured patients' eligibility for benefits update the patient account ledger file a claim with the insurance company...
-
Alimony and separate maintenance payments made under a divorce agreement executed after 2018 are neither taxable to the recipient nor deductible by the payor. What are conditions for divorced to re...
-
How Convertible securities are desirable as financing vehicles because they can be , whereas the the only way to induce the exercise of warrants is to have a provision
-
Your client often comes with different taxation issues. List at least three features of complex taxation issues.
-
Ram Corporation paid cash dividends totaling $55,000 during its most recent fiscal year. How should this information be reported on Ram's statement of cash flows?
-
Late one night, a deputy sheriff found Ronald Rico in his car parked on the side of a road in a rural area of the county. Rico was at the wheel, the headlights were on, and the radio was playing at a...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
Ethylene at 30 bar and 100C passes through a heaterexpander and emerges at 20 bar and 150C. There is no flow of work into or out of the heater-expander, but heat is supplied. Assuming that ethylene...
-
Show the products of thesereactions: CH3 Br CH,CH,OH CH,OH a) Ph- -Br b) Ph CH; CH, Br . CH;OH Br d) ELOH Ph
-
Show the products of thesereactions: Br Br O NaOH NAOH b) PHCHCHCCH3 a)
-
Show the substitution and/or elimination products for these reactions. Explain which mechanism are occurring and which product you expect to be the majorone. OTs a) CH,CH,CHCH, + CH,CH,CO CH,CH,CO,H...
-
Include your strategies for capitalizing on your strengths and overcoming your weaknesses in order to become successful in your work related goals
-
when Recognizing and Identifying Child Abuse and Neglect, Include some strategies for capitalizing on strengths and overcoming weaknesses in order to become successful in your work-related goals?
-
What can be the most beneficial when learning about Recognizing and Identifying Child Abuse and Neglect? How can Recognizing and Identifying Child Abuse and Neglect will assist you in achieving...

Study smarter with the SolutionInn App


