Show the products of thesereactions: Br S t-BUOH a) CH,CH,CH,CH, + t-BUOK + NaOCH,CH, b) Br
Question:
Show the products of thesereactions:
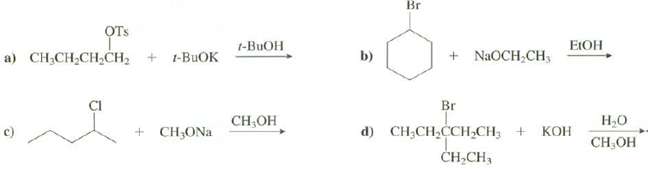
Transcribed Image Text:
Br ОтS t-BUOH a) CH,CH,CH,CH, + t-BUOK + NaOCH,CH, b) ЕОН Br d) CH;CH,CCH,CH3 + ČH,CH, CH-OН Н.о + CH;ONa КОН CH ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (21 reviews)
aCH3C...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Danbury Inc is a resident Canadian corporation with worldwide operations. Canadian operations resulted in taxable income of $ 1 . 1 0 million and non - Canadian operations resulted in taxable income...
-
When analysing cost behaviour, what do we mean by 'level of activity'?
-
Wilcox Automotive Company manufactures an engine designed for motorcycles and markets the product using its own brand name. Although Wilcox has the capacity to produce 50,000 engines annually, it...
-
Calculate the solution to the following SDE: \[d X_{t}=\alpha\left(m-X_{t} ight) d t+\sigma d B_{t}\] with \(X_{0}=x\). The process satisfying this equation is called the meanreverting...
-
Consider the data in Figure for Daisys Dance Studio: Adjustment Data a. Insurance expired, $100. b. Dance supplies on hand, $1,100. c. Depreciation on dance equipment, $2,000. d. Salaries earned by...
-
why can FinTech have important risk management implications for financial institutions?
-
. Consider the below commands i) sed "s/CSH/csh/g" temp > | Is ~/newtemp ii) Is -v grep wc -1 iii) ls -a | grep "." | wc -1 iv) type -l export | type -t Is Which of the above Commands are executed?...
-
Show the products of thesereactions: C, CH3 H,SO4 b) Co + CH,O a) + CH,OH CH, CH, 1) LIAIH4, ether c) 2) ,"
-
Explain whether these elimination reaction would be a good way to prepare thesealkenes: Cl H,O CH,OH + KOH CI EIOH PHCH=CHCH, b) PHCH CHCH; + NaOEt
-
A firm has actual sales of $65,000 in April and $60,000 in May. It expects sales of $70,000 in June and $100,000 in July and in August. Assuming that sales are the only source of cash inflows and...
-
In an ideal diesel cycle, the temperatures at the beginning and at the end of compression are \(57^{\circ} \mathrm{C}\) and \(603^{\circ} \mathrm{C}\), respectively. The temperatures at the beginning...
-
Discuss the assumptions and advantages of multistage compression.
-
A dual cycle operates with a compression ratio \(r_{\mathrm{k}}=10\) and cut-off ratio 1.6. The maximum pressure is given by \(P_{\max }=60 P_{1}\), where \(P_{1}\) is the pressure before...
-
In an air standard diesel cycle, the temperatures at the start and at the end of compression stroke are \(298 \mathrm{~K}\) and \(800 \mathrm{k}\), respectively. The energy added at constant pressure...
-
Explain the working principle of the centrifugal compressor.
-
If the disk in Figure 5.16 rotates in the clockwise direction by \(10^{\circ}\), determine the elastic potential energy of the system. Assume that the springs are originally undeformed. FIGURE 5.16...
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
Find the slope of the line through each pair of points. (18, 13), (14,-5)
-
Propose structures for compounds that fit the following descriptions: (a) An alkyl halide that gives a mixture of three alkenes on E2 reaction (b) An organo halide that will not undergo nucleophilic...
-
Draw all isomers of C4H9Br, name them, and arrange them in order of decreasing reactivity in the SN2 reaction.
-
The following Walden cycle has been carried out. Explain the results, and indicate where Walden inversion isoccurring. OTos H-CH TosCI CHH2 eat CHH2 CHH2 CHCH2 lalp = +33.0 lalp = +31.1 [alp = -19.9...
-
Given the function. f(x) = ln (2x+3x) Compute f'(x). Enter your answer rounded to the nearest tenth
-
A motor-cycle manufacturing company desires a profit of $600,000. The fixed costs are $800,000. The unit selling price of the motor-cycle is $750 and the variable cost per unit is $680. How many...
-
3. ZR Corporation's stock has a beta coefficient equal to 0.8 and a required rate of return equal to 11 percent. If the expected rate of return on the market is 12.5 percent, what is the risk-free...

Study smarter with the SolutionInn App


