Show the products of these reactions and explain whether each would follow an SN1 or an SN2
Question:
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism:
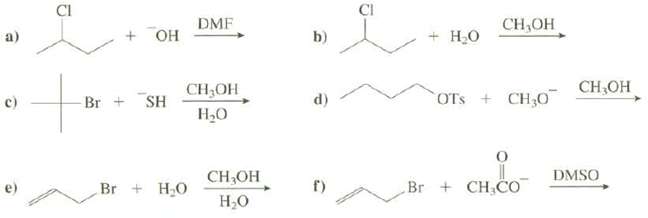
Transcribed Image Text:
a) C) B CI + OH Br + SH DMF CH₂OH H₂O Br + H₂O CH₂OH H₂O b) d) f) CI + H₂O CH,OH OTS + CH₂0 Br + CH₂CO CH₂OH DMSO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a The substrate is a secondary alkyl halide so the mechanism depends on the nucleophile ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain whether these reactions follow an SN1 or an SN2 mechanism. CH3 T a) CH3-C-Br + CHCO CH3 b) OMS d) + SH CH3 c) CH CH-C-0 T CH3 DMF CHCH-Br + CHOH + CH CO + CHCHOH CH,COH 1-BUOH CHOH CHCCH EtOH
-
Show the products of these reactions of 1-propylcyclopentene with thesereagents: a) Br, CCl, b) Br2, H2O d) HBr f) [OsO,], t-BuOOH c) 1) BH3, THF; 2) H,O,, NaOH e) H2O, H,SO, g) 1) O; 2) (CHz)S
-
Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
-
A 15 mm 15 mm silicon chip is mounted such that the edges are flush in a substrate. The chip dissipates 1.4 W of power uniformly, while air at 20C (1 atm) with a velocity of 25 m/s is used to cool...
-
Triangle Zipper Company accumulates costs for its single product using process costing. Direct material is added at the beginning of the production process, and conversion activity occurs uniformly...
-
Barbara Whitley had great expectations about her future as she sat at her graduation ceremony in May 2015. She was about to receive her Master of Accountancy degree, and the following week she would...
-
Items in the table below need to be categorized (if relevant) as part of an income statement. Indicate whether each of the accounts falls under the category of operating expense, cost of goods sold,...
-
What kinds of portable IT help employees work more efficiently and effectively? What may interfere with productivity?
-
Given the logistic model f(x) = Provide your answer below: 800 1+19e-0.402z what is the initial value? Round your answer to the nearest whole number.
-
How would you define and measure customer value to Henkel? Should it define value on the individual level or on the segment level? What is the look and feel of strategic CRM in the FMCG environment?...
-
Arrange these compounds in order of increasing SN1 reaction rate: Ph-Br Br + Br CI
-
Explain whether each pair of reaction should follow an SN1 or an SN2 mechanism. Then explain which member of the pair should proceed at a faster rate. a) b) c) d) f) 09 h) ta -CI+ CHOH OTS T CHCH Br...
-
Explain the relationship between three Statement objects, and illustrate why and how the CallableStatement object can use the setXXX() methods defined in the PreparedStatement interface.
-
Was it difficult to come to consensus on various elements when crafting the culture? What sorts of disagreements arose, and how did you solve them?
-
What results when there is an increase in overall prices? a. Recession b. Deflation c. Peak d. Inflation
-
Are there other strategies that organizations could employ to ensure that underrepresented employees and managers are not disproportionately affected by the layoffs?
-
Take the speed of sound in air at standard temperature and pressure to be \(343 \mathrm{~m} / \mathrm{s}\). Convert this speed to kilometers per hour, inches per nanoseconds, and miles per second.
-
Do you agree with this approach to dismantling the toxic culture? Explain?
-
Design a tower packed with \(50-\mathrm{mm}\) ceramic Hiflow rings for the carbon disulfide scrubber of Problem 5.11. Assume isothermal operation and use a liquid rate of 1.5 times the minimum and a...
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
Solve each system by graphing. y = -x - 2 y =-4.x + 4 -1 ch 2 T 2 ch y + WH x
-
(S)-3-Methylhexane undergoes radical bromination to yield optically inactive 3-hromo-3-methylhexane as the major product. Is the product chiral? What conclusions can you draw about the radical...
-
Assume that you have carried out a radical chlorination reaction on (R)-2-chloro- pentane and have isolated (in low yield) 2, 4-dichioropentane. How many stereoisomers of the product are formed and...
-
What product(s) would you expect from the reaction of 1, 4-hexadiene with NBS? What is the structure of the most stable radical intermediate?
-
Silverton Confectionery is a growing Berkshire-based company specialising in selling quality chocolates and sweets at higher than average prices through newsagents and confectioners. At present their...
-
4. X, the proprietor of a departmental store, decided to calculate separate profits for his two departments L and M for the month ending 31st January. Stock on 31st January could not be valued for...
-
What level of confidentiality should be attached to the preparation and handling of a memorandum of law? Why? Assume you have been working for a legal specialist in estate law for a number of years...

Study smarter with the SolutionInn App


