Show the products of thesereactions: 1) OsO4, ether 2) Na,SO, KMNO4 b) CH,CH;CH=CH2 NaOH CH,CH2 CH3
Question:
Show the products of thesereactions:
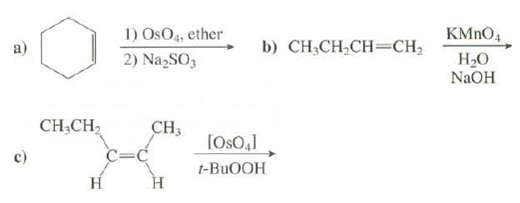
Transcribed Image Text:
1) OsO4, ether 2) Na,SO, KMNO4 b) CH,CH;CH=CH2 Но NaOH CH,CH2 CH3 [Oso,1 c) t-BUOOH н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 82% (17 reviews)
a OH HO b ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
a) What would be the third term? What would be the next to last term? What would be the last term? b) What would be the second term? What would be the next to last term? What would be the last term?...
-
What are the critical success factors for Big Data analytics?
-
NASA trains astronauts to deal with weightlessness (and its associated nausea) by flying them in the "vomit comet," a modified KC-135 airplane that flies in an oscillating path with a period of 72...
-
You are considering an investment in a tree farm. Trees grow each year by the following factors: The price of lumber follows a binomial lattice with \(u=1.20\) and \(d=9\). The interest rate is...
-
A 2.00-kg stone is sliding to the right on a frictionless horizontal surface at 5.00 m/s when it is suddenly struck by an object that exerts a large horizontal force on it for a short period of time....
-
In a statistically based quality improvement project for the turning of steel shafts it was observed that about 23 percent of the shafts failed to meet specifications. Those failures were divided...
-
Harold Co. reported the following current-year purchases and sales data for its only product. Harold uses a perpetual inventory system. Determine the costs assigned to ending inventory and to cost of...
-
Show the products of thesereactions: CH3 . PHCO;H MCPBA NaOH b) C= a) ,
-
Show syntheses of these compounds from(Z)-2-butene: ) - a) H H C-C - CH3 . .
-
A stock sells for $10 per share. You purchase 100 shares for $10 a share (i.e., for $1,000), and after a year the price rises to $17.50. What will be the percentage return on your investment if you...
-
A space vehicle of mass 8,000 kg (including crew) when empty of fuel takes on 42,000 kg of fuel at a space station. It is released from the station with an initial speed of 2 m/s, and its rockets...
-
The 12V battery in your car operates a 25A car stereo. What is the resistance of this stereo system? A "2D" Maglite flashlight runs on 3.0V. What is the current through the bulb if resistance is 15 Q...
-
16. A magnetic field is directed into the page, as shown by the "X" symbols in the small circles. A charged particle, moving in the plane of the page, follows a clockwise spiral of decreasing radius...
-
Some polarized light has an electric field amplitude of 2700 V/m. After it passes through a polarizer, its new electric field amplitude is 1000 V/m. i. The angle (in degrees) between the light's...
-
If a balloon is filled with 932.20 cubic meters of a gas (at 0 o C temperature and 1 Atm pressure). What is the mass of the gas (in kilograms) if its density is 0.08 kilograms per cubic meter? Round...
-
What is the relationship between the MPC and the multiplier? Would a larger value for the MPC tend to increase or decrease the stability of the economy? Briefly explain.
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Calculate the pH of a 0.10-M solution of sodium phosphate. (See Exercise 183.) Data in Exercise 183 Consider the species PO 4 3- , HPO 4 2- , and H 2 PO 4 - . Each ion can act as a base in water....
-
One of the reactions used in determining the sequence of nucleotides in a strand of DNA is reaction with hydrazine. Propose a mechanism for the following reaction, which occurs by an initial...
-
Which compound in each of the following pairs is more basic? (a) CH 3 CH 2 NH 2 or CH 3 CH 2 CONH 2 (b) NaOH or CH 3 NH 2 (c) CH 3 NHCH 3 or pyridine
-
What product would you expect from Hofmann elimination of a heterocyclic amine such as piperidine? Write all thesteps? Piperidine
-
s Machine-hours required to support estimated production Fixed manufacturing overhead cost Variable manufacturing overhead cost per machine-hour Required: 1. Compute the plantwide predetermined...
-
How do visionary leaders navigate the complexities of organizational dynamics to cultivate a culture of innovation and adaptability ? Explain
-
Finnish Corporation manufactures custom molds for use in the extrusion industry. The company allocates manufacturing overhead based on machine hours. Selected data for costs incurred for Job 532 are...

Study smarter with the SolutionInn App


