Show the structures of the likely fragments you would expect in the mass spectra of the followingmolecules:
Question:
Show the structures of the likely fragments you would expect in the mass spectra of the followingmolecules:
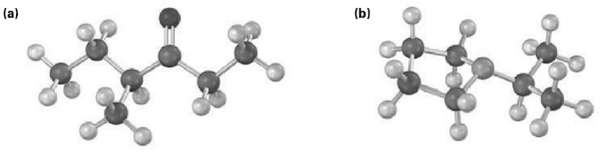
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a The mass spectrum of this ketone shows fragments resulting fro...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
-
Show the structures of the polymers that could be made from the following monomers (yellow-green =Cl): (b) (a)
-
Show the structures of all possible acid-catalyzed dehydration products of the following. If more than one alkene is possible, predict which one will be formed in the largest amount. a....
-
A 25,000 kW turbo-generator is supplied with 128,000 kg/h of steam at 2.50 MPa and 400C when developing it rated load. There are actually extracted 10,400 kg h at 0.3 MPa and 8300 kg/h at 0.06 MPa....
-
Recall from Chapter 2 Adam Smith's dictum, "The division of labor is limited by the extent of the market." How does market growth affect the viability of a focus strategy?
-
What is the relationship among a teams cohesiveness, performance norms, and performance results?
-
Explain why and under what circumstances an employee handbook can be considered a contract.
-
Calculate the child credit for the following taxpayers. Please show your work. a. Jeremy is a single (head of household) father with a dependent 8-year-old son and $79,600 of AGI:...
-
Mason and John operate a computer consulting service as a general partnership. Their partnership agreement states that each partner's share of the profits will be prorated according to the number of...
-
ABC is an electronic manufacturing company based in Singapore. It sells a wide rangeof products to markets in the Asian region. Currently, it has a sizeable IT division withthe following...
-
Where in the IR spectrum would you expect each of the following molecules toabsorb? (a) (c) (b)
-
Propose structures for compounds that fit the following mass-spectral data: (a) A hydrocarbon with M + = 132 (b) A hydrocarbon with M + = 166 (c) A hydrocarbon with M + = 84
-
Following the instructions for drawing the Ï molecular orbital energy levels of the compounds shown in Figure 15.2, draw the Ï molecular orbital energy levels for the cycloheptatrienyl...
-
Explain the advantages and disadvantages of budgetary control (i.e. cash budget, fixed budget, and flexible budget).
-
Taylor's bookkeeper records three items: $5,000 of unearned revenue being earned $1,000 cash collected from earnings recorded in the prior period $3,000 cash collected in advance of services. What is...
-
A company offering short term loans agrees to lend Nicholas $1,200. The amount (plus interest) is repayable in 12 years, and the simple interest rate is 12%. What is the total amount that must be...
-
Mary had a large investment portfolio consisting of equities that she has held for many years. The securities appreciated significantly in value over the years. When she first bought these equities...
-
Allen Corporation reported total sales of $220,000 and total expenses of $180,000 for the year ended December 31, 2023. If the company uses the income summary account for closing entries, what is the...
-
Andrew, a single individual, has $220,000 in salary and $60,000 in self-employment income in 2017. How much must he pay for his additional Medicare surtax?
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
A solution is prepared by dissolving 11.60 g of a mixture of sodium carbonate and sodium bicarbonate in 1.00 L of water. A 300.0 cm 3 sample of the solution is treated with excess HNO 3 and boiled to...
-
Predict the major product(s) of the following reactions: (a) phenylacetylene + 2 HBr (b) hex-1-yne + 2 HBr (c) cyclooctyne + 2 HCl (d) hex-2-yne + 2 HCl + 2 HBr
-
The 2 radical is more stable than 1. The anti-Markovnikov orientation occurs because the bromine radical attacks first to make the most stable radical, in contrast to electrophilic addition where the...
-
Show how hex-1-yne might be converted to (a) 1,2-dichlorohex-1-ene (b) 1-bromohex-1-ene (c) 2-bromohex-1-ene (d) 1,1,2,2-tetrabromohexane (e) 2-bromohexane (f) 2,2-dibromohexane
-
4. Methane burns in air with an equivalence ratio of =0.85, determine the composition of the products: What are the Mass Fractions of each species in the Products?
-
1. Describe the health and safety requirements of the area in which the installation activity is to take place, and the responsibility these requirements place on the learner 2. Describe the...
-
1. Which is easier for nucleation in solids, homogeneous or heterogeneous nucleation? What are the common defects present in solid solution (list at least five kinds of defects)? Are the defects...

Study smarter with the SolutionInn App


