Question: Show the structures of the carbocation intermediates you would expect in the following reactions: (b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
Show the structures of the carbocation intermediates you would expect in the following reactions:
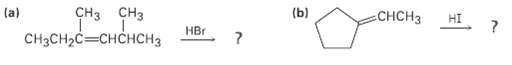
(b) CH3 (a) CH3CH2CCHCH3 HI CH H Er
Step by Step Solution
3.41 Rating (173 Votes )
There are 3 Steps involved in it
The more stable carbocation is formed a ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-OC-A (93).docx
120 KBs Word File


