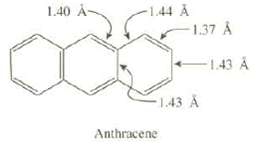
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the
Question:
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the following structure are in accord with predictions based on these resonance structures:
Transcribed Image Text:
1.40 A- -1.44 A -1.37 A -1.43 A Anthracene -1.43 A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The longest bonds 144 and 143 are single in 3 structures ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
For each of the structures below, draw the resonance structure that is indicated by the curved arrows. Be sure to include formal charges. a. b. c. d. e. f. g. h. N-
-
The following two drawings are resonance structures of one compound: But the following two drawings are not resonance structures: They are, in fact, two different compounds. Explain. Not resonance...
-
Cost-Volume-Profit (CVP) analysis can be used to determine the effect of changes in costs and volumes on a company's net profits. This project assignment assumes that you are newly hired as an...
-
Clarke Auto Parts Company manufactures replacement parts for car repairs. The company recently installed a flexible manufacturing system (FMS), which has significantly changed the production process...
-
Is there enough evidence to infer that the incomes (INCOME) of American-born and foreign-born residents differ?
-
In a laboratory test of a new automobile engine design carried out at the Colorado School of Mines, the emission rate (in milligrams per second) of oxides of nitrogen was measured for 32 engines at...
-
The Osborn Construction Company began operations January 2, 2007. During the year, Osborn entered into a contract with Redbeard Razor Corporation to construct a manufacturing facility. At that time,...
-
How do situational factors such as task interdependence, goal clarity, and resource availability influence team coordination, communication patterns, and ultimately, task performance outcomes ?
-
Assume that we make an enhancement to a computer that improves some mode of execution by a factor of 10. Enhanced mode is used 50% of the time, measured as a percentage of the execution time when the...
-
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond of Phenanthrene would your predict to be the shortest? Phenanthrene
-
One general reaction of radicals is the coupling of one with another to form a bond, as shown in the following equation: The following coupling reaction gives two products. Show the structures of...
-
Solve each equation. Vm +3m +12 - m - 2 = 0
-
A composite made of aluminum matrix with steel fibers is designed for carrying electrical power. Also, the electrical resistivity of aluminum is 50 x10^-7 Ohm.cm and that of steel is 400x10^-7...
-
RESUME PROJECT: Develop a detailed job description for Patient Care Associate/ Patient care technician. Compare this description to your current resume. What similarities and differences do you...
-
In Innovation as Usual: How to Help Your People Bring Great Ideas to Life (2013), Miller and Wedell-Wedellsborg discuss the importance of establishing systems within organizations that promote not...
-
Jackson Foundry uses a predetermined overhead allocation rate to allocate overhead to individual jobs, based on the machine hours required.View the costs.Read the requirements.Requirement 1 . Compute...
-
On January 1, 2021, Nash Corp. had 502,000 shares of common stock outstanding. During 2021, it had the following transactions that affected the Common Stock account. February 1 Issued 125,000 shares...
-
Why is it important for strategic planning to begin with a clear sense of the objective?
-
On January 2, 20X3, Sheldon Bass, a professional engineer, moved from Calgary to Edmonton to commence employment with Acco Ltd., a large public corporation. Because of his new employment contract,...
-
(a) Approximate the function f(x) = 3 x by a Taylor polynomial of degree 2 at a = 8. (b) How accurate is this approximation when 7 x 9?
-
Hydrocortisone, a naturally occurring hormone produced in the adrenal glands, is often used to treat inflammation, severe allergies, and numerous other conditions. Is the indicated ?OH group in the...
-
A1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group axial and one group equatorial. Explain.
-
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
-
What is meant by the evolution of the chef with regard to supervisory position?Explain Words count 350 words.
-
Total Direct Materials Variance (b) Total Direct Labor Variance Based on the following, what is the total direct materials variance? Total Product Cost Flexible Budget Variance (a) Total...
-
3 The total force (in N) on the circular end of a water tank is F = 18,600 Sy9-y dy. Evaluate F. 0

Study smarter with the SolutionInn App


