Question: Steady-state evaporation (Fig. 18.2-1), rework the problem solved in S18.2, dealing with the evaporation of liquid A into gas B, starting from Eq. 19.1-17. (a)
Steady-state evaporation (Fig. 18.2-1), rework the problem solved in S18.2, dealing with the evaporation of liquid A into gas B, starting from Eq. 19.1-17.
(a) First obtain an expression for v*, using Eq. (M) of Table 17.8-1, as well as Fick's law in the form of Eq. (D) of Table 17.8-2.
(b) Show that Eq. 19.1-17 then equation: becomes the following nonlinear second-order differentia
(c) Solve this equation to get the mole fraction profile given in Eq. 18.2-11.
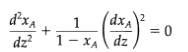
(dxA dxA dz? 1- XA ( dz
Step by Step Solution
3.30 Rating (156 Votes )
There are 3 Steps involved in it
Steadystate evaporation a From Eq M of Table 1781 and the fact that B is stagn... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
6-E-C-E-T-P (298).docx
120 KBs Word File


