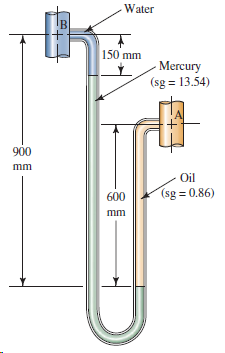
For the manometer shown in Fig. 3.30, calculate (p A - p B ). Water IB 150
Question:
Transcribed Image Text:
Water IB 150 mm - Mercury (sg = 13.54) 900 mm Oil (sg = 0.86) 600 mm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
p B w 15 m m 075 m o 060 ...View the full answer

Answered By

Felix Mucee
I am a detailed and thorough professional writer with 5 years of administrative experience- the last 2 years in academic writing and virtual office environment. I specialize in delivering quality services with respect to strict deadlines and high expectations. I am equipped with a dedicated home office complete with a computer, copier/scanner/fax and color printer.
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past five years. I can bring value to your business and help solve your administrative assistant issues.
4.70+
13+ Reviews
33+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the manometer shown in Fig. 3.28, calculate (p A - p B ). Oil (sg = 0.85) 8 in Water 33 in 12 in
-
For the manometer shown in Fig. 3.29, calculate (p A - p B ). Oil l 150 mm (sg = 0.90) Water 750 mm Mercury (sg = 13.54) 500 mm
-
Calculate the absolute pressure, P1, of the manometer shown in Fig. P1 - 78 in kPa. The local atmospheric pressure is 758 mmHg. Atmosphenc pressure 12 cm 15 cm 3 cm Fluid B 30 cm Fluid A 10 kN/m
-
Assume that when human resource managers are randomly selected, 57% say job applicants should follow up within two weeks. If 9 human resource managers are randomly selected, find the probability that...
-
At December 31, 2016, Durango Ltd. reported the following as plant assets. Land...................................................................................... 2,000,000...
-
A viscously damped spring-mass system is excited by a harmonic force of constant amplitude F0 but varying frequency w. If the amplitude of the steady-state motion is observed to decrease by a factor...
-
Verify that the \(j\)-factor is related to the drag coefficient by the relation \[j_{\mathrm{D}}=\frac{c_{\mathrm{D}}}{2}\] for mass transfer for flow over a flat plate.
-
The model for making tactical decisions described in the text has six steps. These steps are listed, out of order, below. Required: Put the steps in the correct order, starting with the step that...
-
Problem: Module 3 Textbook Problem 5 Learning Objective: 3-6 Using the straight-line method show how bonds issued at a discount. affect financial statements Diaz Company issued $91,000 face value of...
-
TAC Industries Inc. sells heavy equipment to large corporations and federal, state, and local governments. Corporate sales are the result of a competitive bidding process, where TAC competes against...
-
For the differential manometer shown in Fig. 3.27, calculate the pressure difference between points A and B. The specific gravity of the oil is 0.85. 10 in 32 in Oil Water 9 in el
-
For the compound manometer shown in Fig. 3.31, calculate the pressure at point A. Oil (sg = 0.90) Water 125 mm 475 mm 250 mm 50 mm Mercury (sg = 13.54)
-
An object of mass m is at rest at the top of a smooth slope of height h and length L. The coefficient of kinetic friction between the object and the surface, k , is small enough that the object will...
-
Two charged objects 1 and 2 are held a distance \(r\) apart. Object 1 has mass \(m\) and charge \(+2 q\), and object 2 has mass \(2 m\) and charge \(+q\). The objects are released from rest. Assume...
-
Sketch the curves using the equations given in Problems 28-51. \(y^{2}-x^{2}=4\)
-
It really does pay to be a CEO of a Fortune 500 firm, according to the AFLCIO. The average S&P 500 chief executive made $13.1 million in 2016, 347 times more than the average U.S. worker did, up from...
-
Your firm is considering the launch of a new product, the XJ5. The upfront development cost is $12 million, and you expect to earn a cash flow of $3.1 million per year for the next five years. Plot...
-
C. Phillips, a sole trader specialising in material for Asian clothing, has the following purchases and sales for March 2017: Mar 1 Bought from Smith Stores: silk 40, cotton 80. All less 25 per cent...
-
What is Hadoop? What are its primary components, and what does each do?
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
Draw the structure of an alkyne that can be converted into 3-ethylpentane upon hydrogenation. Provide a systematic name for this compound.
-
Propose a mechanism for each of the following transformations: (a) (b) Na NH3 (1) ,
-
Draw the expected product of each of the following reactions, showing stereochemistry where appropriate: . D NANH, NH3
-
Linux - choose how you will manage your software install packages? Analyse some advantages of using a package manager to manage software?
-
A firm produces output according to the production function Q = K1/2 1/2. The demand schedule for the output is P= 20 - Q. If K is fixed at 4 units, what is this firm's short-run demand curve for...
-
Write about the Biden administration's State of the Union address by taking one side and examining how it affects you as an individual. Discuss the policy, economic accomplishments, what he has done...

Study smarter with the SolutionInn App


