Question: For the tank shown in Fig. 3.21, determine the reading of the bottom pressure gage in psig if the top of the tank is vented
For the tank shown in Fig. 3.21, determine the reading of the bottom pressure gage in psig if the top of the tank is vented to the atmosphere and the depth of the oil h is 28.50 ft.
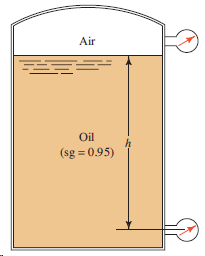
Air Oil (sg = 0.95)
Step by Step Solution
★★★★★
3.36 Rating (171 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

p h 095 624 ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


