Question: Solve for x. Here, we see that we can solve an equation in which the unknown is an element of a determinant. 12 0-1 3
Solve for x. Here, we see that we can solve an equation in which the unknown is an element of a determinant.
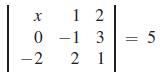
12 0-1 3 -2 2 1 X || 5
Step by Step Solution
3.36 Rating (152 Votes )
There are 3 Steps involved in it
We may leverage the knowledge that a matrixs determinant can be determined by exp... View full answer

Get step-by-step solutions from verified subject matter experts


