Identify the conjugate acids and bases in the following pairs of substances: (a) (CH3)3 NH(CH3) 3N (c)
Question:
Identify the conjugate acids and bases in the following pairs of substances: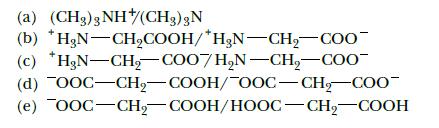
Transcribed Image Text:
(a) (CH3)3 NH(CH3) 3N (c) (b) *HgN-CH₂COOH/*H3N-CH₂-COO H₂N-CH₂-COO7H₂N-CH₂-COO (d) OOC-CH₂-COOH/OOC-CH₂-COO- (e) OOC–CH, COOH/HOOC–CH, COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CH33NH conjugate acid CH33N conjugate base b H3N...View the full answer

Answered By

FREDRICK MUSYOKI
Professional Qualities:
Solution-oriented.
Self-motivated.
Excellent problem-solving and critical thinking skills.
Good organization, time management and prioritization.
Efficient troubleshooting abilities.
Tutoring Qualities:
I appreciate students as individuals.
I am used to tailoring resources for individual needs.
I can integrate IT into student's lessons.
I am good at explaining concepts.
I am able to help students progress.
I have a wide curriculum knowledge.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Biochemistry
ISBN: 9781305961135
9th Edition
Authors: Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Question Posted:
Students also viewed these Sciences questions
-
Identify conjugate acids and bases in the following pairs of substances: (a) (HOCH), CNH (HOCH CNH (b) HOCH, CH, N N CH, CH, SO HOCH, CHN N CH, CH, SOs (c) O SCH, CH, N, N+CH, CH, SOg O SCH, CH,N N...
-
Identify the conjugate acids and bases in the following pairs of substances: (a) (CH 3 ) 3 NH + / (CH 3 ) 3 N (b) + H 3 NCH 2 COOH/ + H 3 NCH 2 COO - (c) + H 3 NCH 2 COO /H 2 NCH 2 COO - (d) OOCCH 2...
-
a. Consider the hydrated aluminum ion Al(H 2 O) 6 3+ as a BrnstedLowry acid. Write the chemical equation in which this ion loses a proton in a reaction with ammonia, NH 3 . Identify the conjugate...
-
Find all values of 0, if 0 is in the interval [0, 360) and has the given function value. cot 0= -1 0= (Type your answer in degrees. Use a comma to separate answers as needed.)
-
1. Suppose a UAWlabor contract with General Dynamics is being renegotiated. Some of the many issues on the table include job security, health benefits, and wages. If you are an executive in charge of...
-
Are there specific decision-making strategies that would be helpful in making an effective decision regarding enrollment management? What communication behaviors would you watch for in the upcoming...
-
What criteria are used to classify species in an evolutionary classification system?
-
At December 31, 2015, Torrealba Company reported the following as plant assets. During 2016, the following selected cash transactions occurred. April 1 Purchased land for $1,200,000. May 1 Sold...
-
How do I design a register in D - ff , T - ff , J - k or SR format? This register can be wrapped to the right by entering a specific command and to the left by entering a second command. Please...
-
Look at Figure 2.17. If you did this titration using TRIS instead of phosphate, how would the titration curve look compared to the figure? Explain. Figure 2.17 Buffering. Acid is added to the two...
-
Both RNA and DNA have negatively charged phosphate groups as part of their structure. Would you expect ions that bind to nucleic acids to be positively or negatively charged? Why?
-
Easy Meals Now (EMN) operates a meal home- delivery service. It has agreements with 20 restaurants to pick up and deliver meals to customers who phone or fax orders to EMN. EMN allocates variable and...
-
An article on the website of the Financial Post, which is published in Canada, discussed the situation of Canadian teams that play in the National Hockey League (NHL). The majority of NHL teams are...
-
Suppose you have a sample of 40 observations and 3 explanatory variables and you want to test for autocorrelation. What can you say about autocorrelation if you have the following DurbinWatson...
-
Obtain a patent of a technology (e.g., go to the patent office Web site) and come up with 10 creative uses of the technology.
-
Inflation, which is an increase in the general level of prices, is generally considered as a very negative phenomenon. Milton Friedman, for example, used to compare it to alcoholism. Therefore, what...
-
In the United Kingdom, patients can access non emergency hospital treatment for free if referred by a family doctor, also known as a general practitioner (GP). A patient gets in contact with the...
-
H and Y Service Station is planning to invest in automatic car wash equipment valued at $250,000. The owner estimates that the equipment will increase annual net cash inflows by $46,000. The...
-
Making use of the tables of atomic masses, find the velocity with which the products of the reaction B10 (n, ) Li7 come apart; the reaction proceeds via interaction of very slow neutrons with...
-
Te amino acid arginine ionizes according to the following scheme: (a) Calculate the isoelectric point of arginine. You can neglect contributions from form I. Why? (b) Calculate the average charge on...
-
It is possible to make a buï¬er that functions well near pH 7 using citric acid, which contains only carboxylate groups. Explain. Citric acid CH CO,H CH2 CO2H
-
It is possible to make a buï¬er that functions well near pH 7 using citric acid, which contains only carboxylate groups. Explain. Citric acid CH CO,H CH2 CO2H
-
The Westerfield Corporation is considering a project with the following cash flows: Time 0: Purchase machinery for $6000 Time 1: Buy inventory $4200 Time 2: Sales (net of expenses) $8000 Time 3:...
-
Explain why the following integral is improper. Then evaluate the improper integral and state whether it converges or diverges. 2x (x + 1)2 dx
-
lpine Inc. uses activity-based costing. The company has two products: A-1 and A-2. Annual production and sales are 3,000 units of A-1 and 2,000 units of A-2. Cost and expected activity information is...

Study smarter with the SolutionInn App


