Look at Figure 2.17. If you did this titration using TRIS instead of phosphate, how would the
Question:
Look at Figure 2.17. If you did this titration using TRIS instead of phosphate, how would the titration curve look compared to the figure? Explain.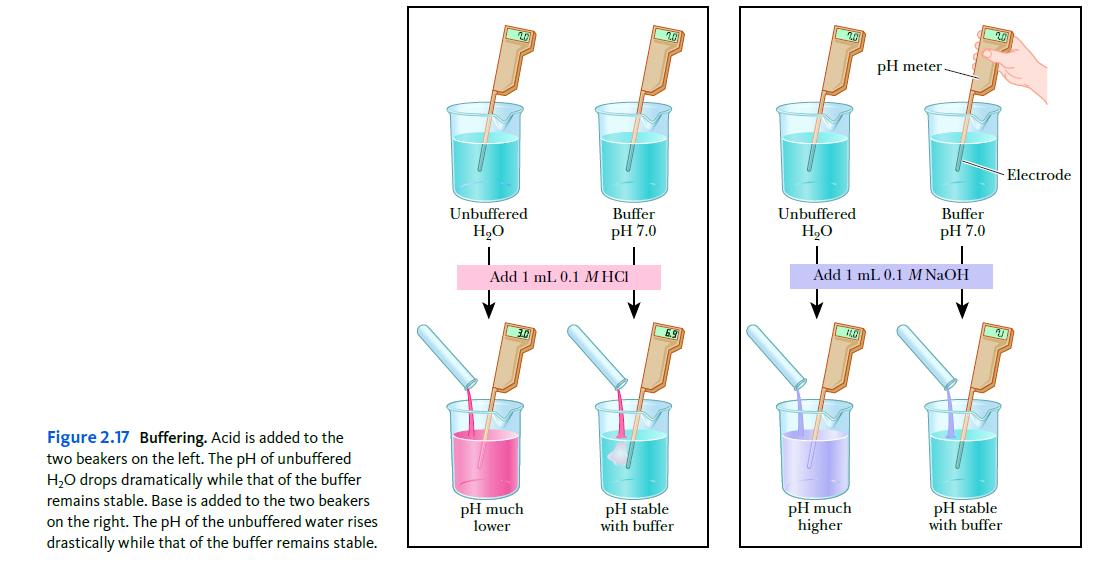
Transcribed Image Text:
Figure 2.17 Buffering. Acid is added to the two beakers on the left. The pH of unbuffered H₂O drops dramatically while that of the buffer remains stable. Base is added to the two beakers on the right. The pH of the unbuffered water rises drastically while that of the buffer remains stable. 20 Unbuffered H₂O Buffer pH 7.0 Add 1 mL 0.1 MHCI pH much lower 201 pH stable with buffer Unbuffered H₂O pH meter pH much higher Buffer pH 7.0 Add 1 mL 0.1 M NaOH - Electrode pH stable with buffer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The titration curve for TRIS ...View the full answer

Answered By

MOHAMMED SIBILI K
I am studying CMA Intermediate at ICMAI and BCOM Degree at IGNOU.
I am completed my higher secondary education (PLUSTWO) at Kerala state board of Higher Secondary Examination, in 2019.
I am completed my high school education (SSLC) at Kerala state board of Examination, in 2017.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Biochemistry
ISBN: 9781305961135
9th Edition
Authors: Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Maria Juarez is a professional tennis player, and your firm manages her money. She has asked you to give her information about what determines the level of various interest rates. Your boss has...
-
The derivative of f(x,y) at Po(1.2) in the direction of 7+is 2/2 and in the direction of -21 it is -3 What is the derivative of f in the direction of -1-21?
-
Based on the best available econometric estimates, the market elasticity of demand for your firms product is 1.50. The marginal cost of producing the product is constant at $75, while average total...
-
What oppressive structures of organization and communication are in evidence in the jobs held by Brandon and Gabriella? Do either Gabriella or Brandon participate in a system of concertive control?...
-
Why are birds considered reptiles in an evolutionary classification system? (a) Feathers evolved from scales. (b) Birds are more similar to reptiles than was previously thought. (c) Birds are...
-
Ron Nord and Lisa Smith are examining the following statement of cash flows for Carpino Company for the year ended January 31, 2010. Ron claims that Carpinos statement of cash flows is an excellent...
-
The following postfix expression with single digit operands is evaluated using a stack: 8 2 3/2 3 * + 5 1 * Note that is the exponentiation operator. The top two elements of the stack after the first...
-
Aspirin is an acid with a pK a of 3.5; its structure includes a carboxyl group. To be absorbed into the bloodstream, it must pass through the membrane lining the stomach and the small intestine....
-
Identify the conjugate acids and bases in the following pairs of substances: (a) (CH3)3 NH(CH3) 3N (c) (b) *HgN-CHCOOH/*H3N-CH-COO HN-CH-COO7HN-CH-COO (d) OOC-CH-COOH/OOC-CH-COO- (e) OOCCH,...
-
1. What was the five-firm concentration ratio in the broadband industry? 2. What would be the revised HHI value if the top dozen firms decided to conduct a horizontal merger and offer a single "Top...
-
Coca-Cola Enterprises is almost 50% debt financed, while Intel, a technology firm, has no net debt. Why might these firms choose such different capital structures?
-
Between 2012 and early 2019, the average rate of inflation as measured by the core personal consumption expenditures (PCE) price index was 1.6 percentbelow the Federal Reserves target of 2 percent....
-
An article in the Wall Street Journal on former Fed Chair Paul Volcker notes that among his bold moves, Mr. Volcker raised the fed-funds rate to an unprecedented 20% to contain double-digit...
-
Dividends per share (DPS), price per share (PPS), and retained earnings (RE) for the 30 Dow Jones industrials for 1984 give us the following multiple regression model: (a) Interpret the multiple...
-
Conduct an Internet search on Senate bill 2232, also known as the Federal Reserve Transparency Act of 2015 and read the contents of the bill. (Note that this particular legislation failed to pass...
-
PPG Industries, founded in 1883, was the first commercially successful plate glass manufacturer in the United States. Today it is a global supplier of coatings, chemicals, and glass. Annually, its...
-
Draw a Feynman diagram for the reaction n + v p + .
-
Carbon dioxide is dissolved in blood (pH 7.4) to form a mixture of carbonic acid and bicarbonate. Neglecting free CO2, what fraction will be present as carbonic acid? Would you expect a signicant...
-
What is the molecular basis for the observation that the overall charge on a protein becomes increasingly positive as pH drops and more negative as pH increases?
-
Te amino acid arginine ionizes according to the following scheme: (a) Calculate the isoelectric point of arginine. You can neglect contributions from form I. Why? (b) Calculate the average charge on...
-
Use the following information to answer the question below: Brazel Corp. has the following information available for the year just ended: Description Amount Net Sales $5,000,000 Cost of Goods Sold...
-
are The graphs of f and f shown. The formula for the 2 function f is f(x) = x +4, x0. Find a formula for f f'(x)= (Type an exact answer, using radicals as needed.) 10- AY 8- y = f(x) 6-
-
Galaxy, Inc. had the following merchandise transactions in October: Purchases $80,000 Purchase returns 8,000 Purchase discounts 7,200 Transportation in 3,000 What is the total cost of merchandise...

Study smarter with the SolutionInn App


