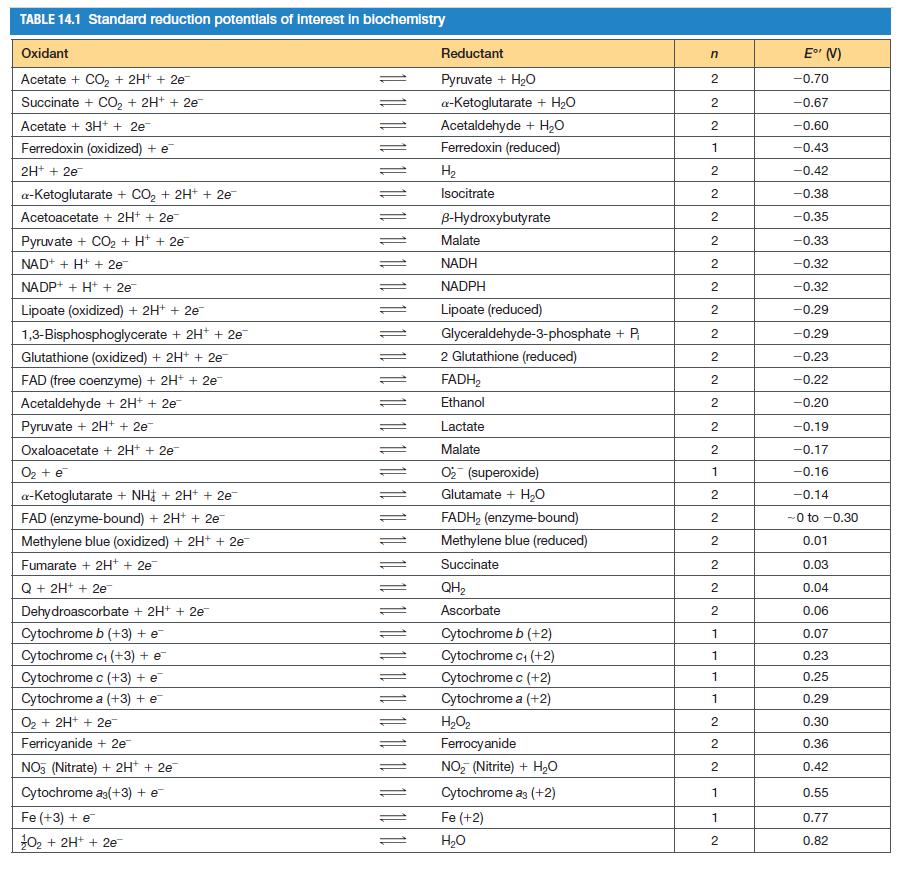
Referring to Table 14.1 for E 0 values, calculate G for oxidation of malate by malate dehydrogenase.
Question:
Referring to Table 14.1 for E′0 values, calculate ∆G°′ for oxidation of malate by malate dehydrogenase.
Table 14.1
Transcribed Image Text:
TABLE 14.1 Standard reduction potentials of interest in biochemistry Oxidant Acetate + CO₂ + 2H+ + 2e¯ Succinate + CO₂ + 2H+ + 2e- Acetate + 3H+ + 2e¯ Ferredoxin (oxidized) + e 2H+ + 2e a-Ketoglutarate + CO₂ + 2H+ + 2e¯ Acetoacetate + 2H+ + 2e™ Pyruvate + CO₂ + H+ + 2e NAD+ + H+ + 2e™ NADP+ +H+ + 2e Lipoate (oxidized) + 2H+ + 2e- 1,3-Bisphosphoglycerate + 2H+ + 2e™ Glutathione (oxidized) + 2H+ + 2e FAD (free coenzyme) + 2H+ + 2e™ Acetaldehyde + 2H+ + 2e Pyruvate + 2H+ + 2e Oxaloacetate + 2H+ + 2e O₂ + e a-Ketoglutarate + NH + 2H+ + 2e FAD (enzyme-bound) + 2H+ + 2e™ Methylene blue (oxidized) + 2H+ + 2e Fumarate + 2H+ + 2e¯ Q + 2H+ + 2e Dehydroascorbate + 2H+ + 2e Cytochrome b (+3) + e Cytochrome c₁ (+3) + e Cytochrome c (+3) + e Cytochrome a (+3) + e O₂ + 2H+ + 2e™ Ferricyanide + 2e™ NO3 (Nitrate) + 2H+ + 2e Cytochrome ag(+3) + e Fe (+3) + e ¹0₂ + 2H+ + 2e = Reductant Pyruvate + H₂O a-ketoglutarate + H₂O Acetaldehyde + H₂O Ferredoxin (reduced) H₂ Isocitrate B-Hydroxybutyrate Malate NADH NADPH Lipoate (reduced) Glyceraldehyde-3-phosphate + P₁ 2 Glutathione (reduced) FADH₂ Ethanol Lactate Malate O₂ (superoxide) Glutamate + H₂O FADH₂ (enzyme-bound) Methylene blue (reduced) Succinate QH₂ Ascorbate Cytochrome b (+2) Cytochrome c₁ (+2) Cytochrome c (+2) Cytochrome a (+2) H₂O₂ Ferrocyanide NO₂ (Nitrite) + H₂O Cytochrome a3 (+2) Fe (+2) H₂O n 2 2 2 1 2 NN 2 2 2 2 2 2 2 NN 2 2 2 2 2 1 2 2 2 2 2 2 1 1 1 1 2 2 2 1 1 2 E° (V) -0.70 -0.67 -0.60 -0.43 -0.42 -0.38 -0.35 -0.33 -0.32 -0.32 -0.29 -0.29 -0.23 -0.22 -0.20 -0.19 -0.17 -0.16 -0.14 -0 to -0.30 0.01 0.03 0.04 0.06 0.07 0.23 0.25 0.29 0.30 0.36 0.42 0.55 0.77 0.82
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To calculate G for the oxidation of malate by malate dehydrogenase you can use the Ner...View the full answer

Answered By

Dorcas Juliet
I am a proficient tutor and writer with over 4 years experience, I can deliver A+ works in all fields related to business and economics subject. Kindly hire me for excellent papers
4.70+
10+ Reviews
51+ Question Solved
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:
Students also viewed these Sciences questions
-
-hydroxybutyrate dehydrogenase catalyzes the last step in ketogenesis, the reduction of acetoacetate to b-hydroxybutyrate. (a) Write a balanced equation for this reaction. (b) Calculate the standard...
-
Freshly prepared mitochondria were incubated with b -hyroxybutyrate, oxidized cytochrome c, ADP, Pi, and cyanide. b -Hyroxybutyrate is oxidized by an NAD+ -dependent dehydrogenase. The experimenter...
-
GSSG + NADPH + H+ 2GSH + NADP+ (a) Calculate G for the glutathione reductase reaction in the direction shown, using E values from Table 14.1. (b) Suppose that a cell contained an isoform of...
-
In general, algorithms are classified into paradigms like: greedy, dynamic, optimization, brute-force, and divide & conquer. Each of the following phrases describes one of these paradigm 1) A...
-
Mags Pie Shop is a rapidly growing baker and distributor of specialty pies for festive occasions. Mags CPA adviser keeps recommending that Mag install better internal controls over the business....
-
On March 1, 20Y1, Sadie May established Pampered Pet, which completed the following transactions during the month: a. Sadie May transferred cash from a personal bank account to an account to be used...
-
This assignment begins with a completed simulation of the toluene hydrodealkylation process in Figure 17.1 and involves the completion of an economic evaluation. Note that the simulation results for...
-
Lidge Company of Texas (TX) is classified as a monthly depositor and pays its employees monthly. The following payroll information is for the second quarter of 2 WITHHOLDINGS Wages $86,100 92,500...
-
Solve the equation below: 4 x ^ 2 = 9
-
Construct a table that lists all of the metabolic reactions from Chapters 12 and 13 that form or break CC bonds. Arrange them according to whether the CC bond is a or b to a carbonyl. Which reactions...
-
Write a balanced equation for the conversion in the glyoxylate cycle of two acetyl units, as acetyl-CoA, to oxaloacetate.
-
A U.K.-based travel agent, Euan Morton, LLC, would like your systems team to design a commandlanguage interface he can use to book seats for airlines to which his firm has solid business ties, such...
-
In an economy where the 2-year spot rate is 3.00%/year and the 6-year spot rate is 5.00%/year what is the 4-year rate 2-years forward? Assume annual compounding.
-
4. Suppose that you estimate the following cost function for your company, which is a firm operating in a perfectly competitive market: TC=100Q-4Q 2 +Q 3 The market demand and supply equations are as...
-
Rebecca is an 18-year-old female high school student set to graduate soon. She is preparing to go to college next fall, and purchasing her first car is one of the first steps toward some really big,...
-
Kelly Sassin and Sarah Conner are working on the following problem: There is a solid, insulating sphere (R =3 m) with a total charge of -20 C and with uniform charge density. What is the electric...
-
15. You would like to have a balance of $600,000 at the end of 15 years from monthly savings of $900. If your returns are compounded monthly, what is the APR you need to meet your goal? Enter your...
-
What are the two basic economic concepts of income? What implications do they have for analysis? Discuss.
-
Read the case study Richter: Information Technology at Hungarys Largest Pharma and answer the following question: How does the organization ensure the accuracy of the data it stores?
-
Organize the following words into two related groups: catabolism, energy-requiring, reductive, anabolism, oxidative, energy-yielding.
-
If a reaction can be written A B, and the G' is 20 kJ mol -1 , what would the substrate/product ratio have to be for the reaction to be thermodynamically favorable?
-
Can you use the equation G' = -2RT ln K' eq to get the G' from the information in Question 3? Question 3 Consider the reaction The K eq at pH 8.5 and 38C is 122. Can you determine the rate of the...
-
How would the addition of 2 south - facing windows that accept sunlight affect the heating requirements on a sunny day. Consider a simple example where both windows are 1 - meter on a side, are made...
-
Along the edge of this page is a scale that you can use to measure angular diameters up to 30 degrees. While standing 200 cm away from a wall that has a paper plate hanging at eye level, hold the...
-
A tugboat tows a ship with a constant force of magnitude F. The increase in the ship's speed during a 10 s interval is 3.0 km/h. When a second tugboat applies an additional constant force of...

Study smarter with the SolutionInn App


