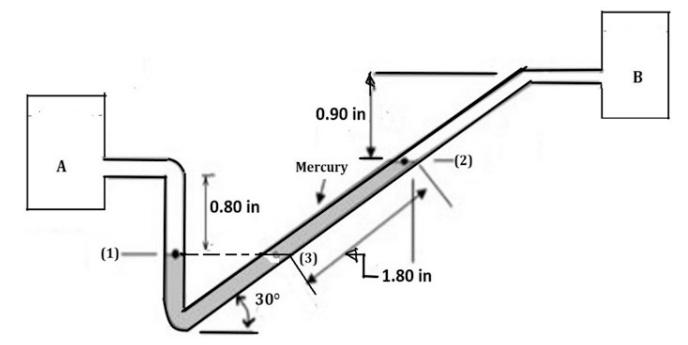
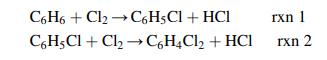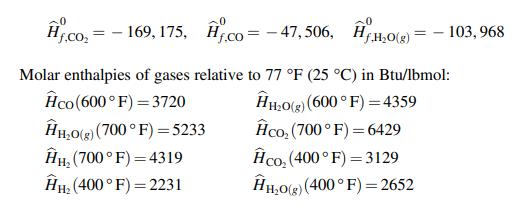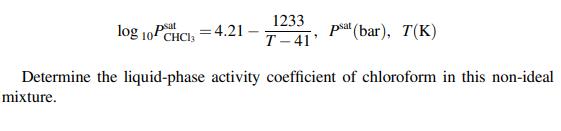Chemical Engineering Principles And Applications 1st Edition Nuggenhalli S. Nandagopal - Solutions
Dive into the comprehensive world of "Chemical Engineering Principles And Applications" by Nuggenhalli S. Nandagopal with our expertly curated resources. Access a vast collection of questions and answers that are designed to enhance your understanding of the textbook's complex concepts. Our online platform offers a detailed solution manual and solutions PDF, providing step-by-step answers to solved problems. Ideal for students and instructors alike, the instructor manual and test bank offer chapter solutions that are perfect for exam preparation. Enjoy the convenience of a free download, ensuring you have all the necessary tools for success in chemical engineering at your fingertips.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()