Let T(x, y) be the temperature at location (x, y) on a thin sheet of metal. Assume
Question:
Let T(x, y) be the temperature at location (x, y) on a thin sheet of metal. Assume that ∇T = (y − 4, x + 2y). Let r(t) = (t2, t)be a path on the sheet. Find the values of t such that
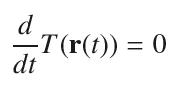
Transcribed Image Text:
d dt T(r(t)) = 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
By the Chain Rule for Paths we have dT rt VTrat r t dt We comput...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
A gas mixture is found to contain two diatomic A and B species for which the partial pressures of both are 0.05065 MPa (0.5 atm). This mixture is to be enriched in the partial pressure of the A...
-
It is desired to enrich the partial pressure of hydrogen in a hydrogen-nitrogen gas mixture for which the partial pressures of both gases are 0.1013 MPa (1 atm). It has been proposed to accomplish...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Problem 3.3 Minimize the functional J(x()) = f sx (s) x (s) ds, = 1 sr (s) i (s) ds, 0 subject to the endpoint conditions x (0) = 0 and x (1) = 1.
-
Methanol is formed from carbon monoxide and hydrogen in the gas-phase reaction The mole fractions of the reactive species at equilibrium satisfy the relation where P is the total pressure (atm). K e...
-
University Food Systems, Inc., has issued a 40 percent stock dividend. The company has 752,000 shares authorized and 200,000 shares outstanding. The par value of the stock is $10 per share, and the...
-
Natural gas ( \(ho=0.0044\) slugs \(/ \mathrm{ft}^{3}\) and \(\left.u=5.2 \times 10^{-5} \mathrm{ft}^{2} / \mathrm{s} ight)\) is pumped through a horizontal 6-in.-diameter cast-iron pipe at a rate of...
-
The unadjusted trial balance of LaBarbara Data at December 31, 2014, appears below. Adjustments had been made until November, but no adjustments for December have been made. Adjustment data: a....
-
Explain some of the limitations placed on these rights by citing at least three instances where this has happened. This means finding three cases where the Bill of Rights protections have been...
-
Find a vector normal to the surface x 2 + y 2 z 2 = 6 at P = (3, 1, 2).
-
Let (x, y, z) = sin(xy + z) and P = (0, 1, ). Calculate D u (P), where u is a unit vector making an angle = 30 with P .
-
Using the ideas discussed below, how would you summarise organisational fluidity? The concept of the virtual organisation appears suited to knowledge-based work that utilises technologies such as the...
-
The issue is whether the order of the trial court was an exercise of its in rem or in persona jurisdiction. The order, entered in the context of the parties' divorce action, directed the parties to...
-
The Mississippi Department of Human Services ("DHS"), on behalf of Diana Goodin-McKay ("McKay"), sought to enforce an Arizona child support order against John T. Goodin ("Goodin") in the Chancery...
-
Since the United States Supreme Court's decision in Zablocki, some commentators have suggested that all restrictions on an individual's right to marry are unconstitutional. Do you think states should...
-
In this dispute over occupancy rights to a rent-controlled apartment, the central question to be resolved on this request for preliminary injunctive relief . . . is whether appellant has demonstrated...
-
Plaintiff avers that in October of 1964 she and defendant "entered into an oral agreement" that while "the parties lived together they would combine their efforts and earnings and would share equally...
-
a. Suppose in problem that a sales tax of $2 per pound is imposed on catnip. What is the new market price of catnip? What price do demanders actually pay? What is the new equilibrium quantity? b....
-
Consider the following cash flows in Table P5.5. (a) Calculate the payback period for each project. (b) Determine whether it is meaningful to calculate a payback period for project D. (c) Assuming...
-
How many two-letter codes can be formed using the letters A, B, C, D, and E? Repeated letters are allowed.
-
How many three-digit numbers can be formed using the digits 0 and 1? Repeated digits are allowed.
-
How many three-digit numbers can be formed using the digits 0, 1, 2, 3, 4, 5, 6, 7, 8, and 9? Repeated digits are allowed.
-
What would be some financial, political and social barriers that a nurse manager may encounter when proposing a team based approach to retain new grad rn's and what are some effective ways to address...
-
Explain in detail. 1. How does an American gain a sense of security/safety with gun ownership, laws and control. 2. How does the sense of protection with a firearm cause a sense of security?
-
Gabbe Industries is a division of a major corporation. Last year the division had total sales of $27,615,900, net operating Income of $3,866,226, and average operating assets of $7,081,000. The...

Study smarter with the SolutionInn App


