The power (in microwatts) of a laser is measured as a function of current (in milliamps). Find
Question:
The power (in microwatts) of a laser is measured as a function of current (in milliamps). Find the linear least-squares fit (Exercise 60) for the data points.
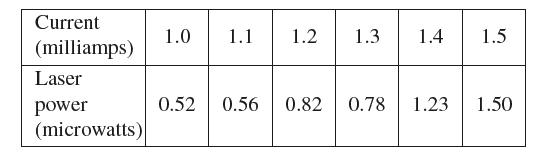
Data From Exercise 60
Given n data points (x1, y1), . . . , (xn, yn), the linear least-squares fit is the linear function

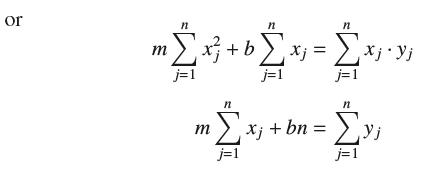
Transcribed Image Text:
Current (milliamps) Laser power (microwatts) 1.0 1.1 1.2 1.3 1.4 0.52 0.56 0.82 1.5 0.78 1.23 1.50
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
Find the best linear least squares fit of the following data using the indicated weights: (a) (b) (c) (d) .2 4 .7 1.2 21 02 yi -5 -3 2 0 3 .5 2 y 2 13 8 2
-
The following table lists values for three variables measured for 60 consecutive eruptions of the geyser Old Faithful in Yellowstone National Park. They are the duration of the eruption (x 1 ), the...
-
When a block of mass M, connected to the end of a spring of mass ms = 7.40 g and force constant k, is set into simple harmonic motion, the period of its motion is T = 2π √M + (ms/3)/k A...
-
What is the best way to describe automation? developing highly advanced robots that can mimic higher-levelhuman thinking making human workers fully reliant on technology to performtheir job...
-
Refer to the data in BE6-7. Assuming quarterly compounding of amounts invested at 12%, how much of Jack Thompsons inheritance must be invested to have enough at retirement to buy the boat?
-
The following labor variance analysis was performed for Longman. Requirements 1. Record Longmans direct labor journal entries. 2. Explain what management will do with this varianceinformation. Actual...
-
The helium-filled balloon shown in Fig P9.81 is to be used as a wind-speed indicator. The specific weight of the helium is \(y=0.011 \mathrm{lb} / \mathrm{ft}^{3}\), the weight of the balloon...
-
On January 1, the Voluntary Action Agency received a cash contribution of $300,000 restricted to the purchase of buses to be used in transporting senior citizens. On January 2 of that same year,...
-
A 1.3 kg book is lying on a 0.75 m -high table. You pick it up and place it on a bookshelf 2.3 m above the floor. During this process, how much work does gravity do on the book? During this process,...
-
Let A = (a, b) be a fixed point in the plane, and let A (P) be the distance from A to the point P = (x, y). For P A, let e A P be the unit vector pointing from A to P (Figure 27): y Distance f(x,...
-
Find the maximum volume of a cylindrical can such that the sum of its height and its circumference is 120 cm.
-
If a default occurs in a non-recourse commercial mortgage-backed security, the lender will most likely: A. Recover prepayment penalty points paid by the borrower to offset losses. B. Use only the...
-
The Rydberg equation gives us the mathematical relationship between: A) an explanation for the continuous range of energy values associated with atomic spectra B) wavelength and frequency C) electron...
-
18. If 5.0 moles of calcium reacts with excess oxygen as shown, 2 Ca(s) + O2(g) 2CaO(s) AHxn= -1,270 kJ a. The reaction releases 6,350 kJ. b. The reaction absorbs 6,350 kJ. c. The reaction releases...
-
When aluminum is placed in concentrated hydrochloric acid, hydrogen gas is produced. 2 Al(s) + 6 HCl(aq) ->> 2 AICI, (aq) + 3 H(g) What volume of H(g) is produced when 7.20 g Al(s) reacts at STP?...
-
Draw a roadmap and write a plan to find the mass ( g ) of the products based on the given amount of reactants. 2 H 2 O 2 - > 2 H 2 O + O 2 given: H 2 O 2 = 4 9 . 4 mg
-
Calculation: In lab, you are given a protein stock that is 7 mg/ml. You want to add 60 ug of this protein to your reaction tube. What volume do you need to pipette of the stock into your reaction...
-
Fall-Line, Inc. is a Great Falls, Montana, manufacturer of a variety of downhill skis. Fall-line is considering four locations for a new plant: As Pen Colorado; Medicine Lodge Kansas; Broken Bow,...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
A function is defined over an interval [a, b]. (a) Graph indicating the area A under from a to b. (b) Approximate the area A by partitioning into four subintervals of equal length and choosing u as...
-
A function is defined over an interval [a, b]. (a) Graph indicating the area A under from a to b. (b) Approximate the area A by partitioning into four subintervals of equal length and choosing u as...
-
A function is defined over an interval [a, b]. (a) Graph indicating the area A under from a to b. (b) Approximate the area A by partitioning into four subintervals of equal length and choosing u as...
-
What are the implications of adopting a holacratic organizational structure for traditional management roles and hierarchies, and how might this influence corporate culture and employee...
-
Pin Corporation, a U.S. company, formed a British subsidiary on January 1, 2023 by investing 450,000 British pounds () in exchange for all of the subsidiary's common stock. The British subsidiary,...
-
A non dividend paying stock is trading at $45. What arbitrage opportunity is present if an American call option on the stock with an exercise price of $42 is trading at $2?

Study smarter with the SolutionInn App


