a. Copy out and complete the table, which covers the different methods of preparing salts. b. Write
Question:
a. Copy out and complete the table, which covers the different methods of preparing salts.
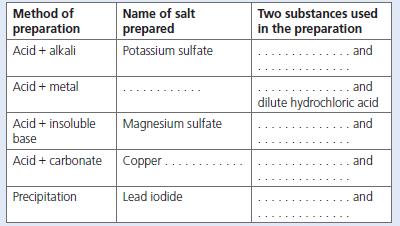
b. Write word and balanced chemical equations for each reaction shown in your table. Also write ionic equations where appropriate.
Name of salt prepared Two substances used in the preparation Method of preparation Acid + alkali Potassium sulfate and Acid + metal .and dilute hydrochloric acid Acid + insoluble base Magnesium sulfate and Acid + carbonate Copper. and Precipitation Lead iodide and
Step by Step Answer:



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
Write the balanced chemical equations for (a) The complete combustion of acetic acid (CH3COOH), the main active ingredient in vinegar (b) The decomposition of solid calcium hydroxide into solid...
-
For each reaction shown here, identify the nucleophile, its atom, the eletrophilic atom in the substate molecule, and the leaving groups. Write the organic product of the reaction. Br + NaSH (a) CHl...
-
Write the balanced molecular and net ionic equations for the reaction that occurs when the contents of the two beakers are added together. What colors represent the spectator ions in each reaction?...
-
Which of the following expressions correctly returns an integer that represents the month of a Local Date object named hireDate? a. GetMonth(hireDate) b. GetMonthValue(hireDate) c....
-
The pH of a cup of coffee (at 25C) was found to be 5.12. What is the hydronium-ion concentration?
-
Interpret the following Excel output. Discuss the structure of the experimental design and any significant effects. Alpha is .05. ANOVA: Two-Factor WithReplication ANOVA Source of Variation SS df MS...
-
Figure \(\mathrm{P} 27. 39\) shows five objects, all placed in the same uniform, upward-directed external magnetic field. Rank the objects according to the amount of magnetic flux through them,...
-
The following data were taken from the financial statements of Hunter Inc. for December 31, 2014 and 2013: The income before income tax was $480,000 and $420,000 for the years 2014 and 2013,...
-
(a) Explain the following methods of payment that could be used by a multinational corporation (MNC) to settle international transactions: Required: (i) Prepayment. (2 marks) (ii) Letters of credit...
-
A study is made by the recording industry in the United States of the number of music CDs owned by 25 senior citizens and 30 young adults. The information is reported below. a. Find the median and...
-
Study the following scheme. a. Give the names and formulae of substances A to E. b. Describe a test which could be used to identify the presence of water. c. Which indicator is suitable for the...
-
Explain, with the aid of examples, what you understand by the following terms: a. Strong acid b. Weak acid c. Strong alkali d. Weak alkali e. Concentrated acid.
-
1. How does the current change in a circuit if the resistance increases by a factor of 2? 2. How does the current change in a circuit if the voltage is increased by a factor of 2? 3. How would the...
-
Anna Goldsworthy is the chief financial officer of a manufacturing firm headquartered in the United Kingdom. She is responsible for overseeing exposure to price risk in both the commodity and...
-
Instructions Your boss has tasked you with installing and maintaining an operating system for a new employee, You will be re-using a computer formerly used by someone who just quit; therefore, you...
-
Explain the following six (6) operating systems: Batch OS Distributed OS Multitasking OS Network OS Real-OS Mobile OS
-
Outline four steps for submitting batch jobs under Linus operating system with specific reference to the iceberg cluster using qsub command.?
-
Given the equation below: Evaluate 100 000 n+n+1 n=1 1.1.1. using a for loop and find the computing runtime 1.1.2. using the vectorising and find the computing the runtime. 1.1.3. What difference in...
-
(a) Show that the nonsingular system ax + by = p, ex -I- dy = q has the solution given by the determinantal ratios Where (b) Use Cramer's Rule (1.87) to solve the systems (i) x + 3y=13. 4x + 2y = 0...
-
Linda Lopez opened a beauty studio, Lindas Salon, on January 2, 2011. The salon also sells beauty supplies. In January 2012, Lopez realized she had never filed any tax reports for her business and...
-
The temperature dependence of the vapour pressure of solid sulfur dioxide can be approximately represented by the relation log (p/Torr) = 10.5916 - 1871.2/ (T/K) and that of liquid sulfur dioxide by...
-
The temperature dependence of the vapour pressure of solid sulfur dioxide can be approximately represented by the relation log (p/Torr) = 10.5916 - 1871.2/ (T/K) and that of liquid sulfur dioxide by...
-
Glucose and fructose are simple sugars with the molecular formula C6HI206 Sucrose, or table sugar, is a complex sugar with molecular formula C12H220I1 that consists of a glucose unit covalently bound...
-
The capital asset pricing model (CAPM) postulates that the mean return of each security (Y) should be linearly related to its Beta (X2), a measure of systematic risk. We examine this hypothesis by...
-
I made some purchases yesterday at Target. I went to check my bank account and I see on my current my credit card statement the charge was $75.43. If sales tax is 8% in my state, what was the total...
-
Discuss the impact of disruptive technologies on business management practices.

Study smarter with the SolutionInn App


