Study the following scheme. a. Give the names and formulae of substances A to E. b. Describe
Question:
Study the following scheme.
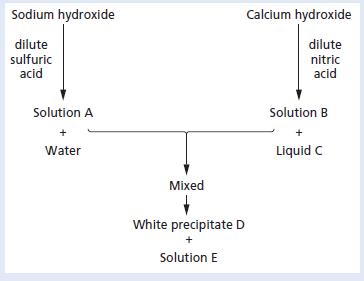
a. Give the names and formulae of substances A to E.
b. Describe a test which could be used to identify the presence of water.
c. Which indicator is suitable for the initial reaction between the hydroxides and the dilute acids shown?
d. Write balanced chemical equations for the reactions taking place in the scheme.
e. Write an ionic equation for the production of the white precipitate D.
Sodium hydroxide Calcium hydroxide dilute sulfuric acid dilute nitric acid Solution A Solution B Water Liquid C Mixed White precipitate D Solution E
Step by Step Answer:
a Substance A Sodium Hydroxide NaOH Substance B Sulphuric Acid H2SO4 Substance C Pot...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
The equation for the initial reaction between an alcohol (primary or secondary) and iodine solution in the presence of aqueous sodium hydroxide is: RCH(OH)R'() + I2(aq) + 2NaOH(aq) RCOR'(aq) +...
-
Write balanced chemical equations for each of the following reactions: (a) The nitric oxide molecule undergoes photodissociation in the upper atmosphere. (b) The nitric oxide molecule undergoes...
-
a. Give the names and formulae of the two major greenhouse gases. b. Name a natural source of the gases you have named in part a. c. Name a man-made source of the gases you have named in part a.
-
This is a stocklist case arising under 220(b) of our [Delaware] General Corporation Law. The issue is whether a shareholder states a proper purpose for inspection under our statute in seeking to...
-
A wine was tested for acidity, and its pH was found to be 3.85 at 25C. What is the hydronium-ion concentration?
-
Are some unskilled office jobs viewed as having more status than others? Suppose a study is conducted in which eight unskilled, unemployed people are interviewed. The people are asked to rate each of...
-
A square loop of side length \(100 \mathrm{~mm}\) is placed on a wooden table in a uniform magnetic field of magnitude \(0.25 \mathrm{~T}\). The greatest magnetic flux through the loop is measured...
-
Dynamo Manufacturing paid cash to acquire the assets of an existing company. Among the assets acquired were the following items: Patent with 4 remaining years of legal life .....$40,000 Goodwill...
-
QUESTION TWO a) State and explain five monetary policy instruments used by the central bank to regulate interest rate, inflation rate, exchange rate and the quantity of money in the economy [15...
-
A differential amplifier has an open-loop voltage gain of 120 and a common input signal of 3.0 V to both terminals. An output signal of 24 mV results. Calculate the common-mode gain and the CMRR.
-
In a titration involving 24.0 cm 3 potassium hydroxide solution against a solution containing 1 mol dm 3 of sulfuric acid, 28.0 cm 3 of the acid was found to just neutralise the alkali completely. a....
-
a. Copy out and complete the table, which covers the different methods of preparing salts. b. Write word and balanced chemical equations for each reaction shown in your table. Also write ionic...
-
Find the value of t for the difference between two means based on an assumption of normality and this information about two samples: Mean 38.2 43.1 Std. Dev. 14.2 10.6 Sample Number 2 2 25
-
Find the present value of $30,000 due in 7 years at the given rate of interest. (Use a 365-day year. Round your answer to the nearest cent.) 3%/year compounded quarterly tA
-
1. A ladder 7 m long is leaning against a wall. If the bottom of the ladder is pushed horizontally toward the wall at 1.5 m/sec, how fast is the top of the ladder sliding up the wall when the bottom...
-
(9%) Problem 11: Two ice skaters, each with the same mass, m, skate in opposite directions at the same speed of v. For the purposes of this problem, treat each skater as a point mass. Their...
-
For each of the following resident individual taxpayers, calculate the amount that they would be entitled to claim under either s.US-6Oor s.25-65 as a deduction for election expenses forthe2O2J/22...
-
What is wrong with this piece of code? Is it stylistic or substantial? char s[MAXARR]; n=0; while (c=getchar () !=-1) s [n++]=c;
-
(a) Show that if is a block diagonal matrix, where A and B are square matrices, then det D = det A det B. (b) Prove that the same holds for a block upper triangular matrix (c) Use this method to...
-
Eleni Cabinet Company sold 2,200 cabinets during 2011 at $160 per cabinet. Its beginning inventory on January 1 was 130 cabinets at $56. Purchases made during the year were as follows: February . 225...
-
A gas obeys the equation of state Vm = RT/p + aT2 and its constant pressure heat capacity is given by Cp,m = A + BT + Cp, where a, A, B, and Care constants independent of T and p. Obtain expressions...
-
What fraction of the enthalpy of vaporization of ethanol is spent on expanding its vapour?
-
There are no dietary recommendations for consumption of carbohydrates. Some nutritionists recommend diets that are largely devoid of carbohydrates, with most of the energy needs being met by fats....
-
C. Preparation of cash budgets for November and December Weller Industrial Gas Corporation Schedule for Expected Cash Disbursements for November and December November December Accounts Payable,...
-
Crazy Crab Cosmetics Company Year 1 Year 2 EBITDA $21,360 $24,750 Total value of equity $243,000 $225,000 Total firm value $364,500 $405,000 In Year 1, Crazy Crab has an entity multiple of (Note:...
-
What is the "Consumer Behavior and Marketing Mix" and why is it so important in strategy formulation and implementation?

Study smarter with the SolutionInn App


