The apparatus shown below was set up. Give explanations for the following observations. a. The formation of
Question:
The apparatus shown below was set up.
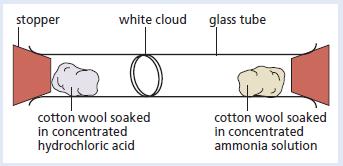
Give explanations for the following observations.
a. The formation of a white cloud.
b. It took a few minutes before the white cloud formed.
c. The white cloud formed further from the cotton wool soaked in ammonia.
d. Cooling the concentrated ammonia and hydrochloric acid before carrying out the experiment increased the time taken for the white cloud to form.
Transcribed Image Text:
stopper white cloud glass tube cotton wool soaked in concentrated hydrochloric acid cotton wool soaked in concentrated ammonia solution
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a the Vapours from the two gases diffuse towards each other and where they meet the...View the full answer

Answered By

Amruta Hajare
I have bachelors degree in general science commonly known as BSC. After teaching vocational colleges for some years I realized my calling to teach was for young minds, giving them strong foundation for future careers and then went back to University and did post graduate diploma in education (PGDE). I am therefore experienced tutor tackling various topics in Biology, chemistry and mathematics under IGCSE curriculum. I give services to my clients at a fair price and relate very well with both parents and students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Concentrated hydrochloric acid contains 1.00 mol HCl dissolved in 3.31 mol H2O. What is the mole fraction of HCl in concentrated hydrochloric acid? What is the molal concentration of HCl?
-
A barium mineral was dissolved in hydrochloric acid to give a solution of barium ion. An excess of potassium sulfate was added to 50.0 mL of the solution, and 1.128 g of barium sulfate precipitate...
-
Concentrated hydrochloric acid is usually available at a concentration of 37.7 percent by mass. What is its molar concentration? (The density of the solution is 1.19 g/mL.)
-
In Exercises find the derivative of the function. y = x(x + 1)
-
A weak acid, HA, is dissolved in water. Which one of the following beakers represents the resulting solution? (Water molecules have been omitted for clarity.)
-
Judges should start offering punitive awards in breachof-contract cases. Punitive damages are awarded for the sole purpose of deterring the defendant and others from doing the same act again. Our...
-
Why would two objects made of nonconducting material make a poorer capacitor than two objects of the same shape made of material that is an electrical conductor?
-
Shirley Hopkins is developing a program in leadership training for middle-level managers. Shirley has listed a number of activities that must be completed before a training program of this nature...
-
Kazibwe, a businessman does not maintain a complete set of books for his business transactions. The following is a summary of his cash transactions for the year ended December 31, 2015. Receipts Kshs...
-
The acceleration ac 5 m/s2 is in the direction shown. From the velocity analysis, it was found that the angular velocity of members AB and BC are respectively @AB= 15 rad/s and @BC = 25 rad/s....
-
Define the following terms using specific examples to help with your explanation: a. Element b. Metal c. Non-metal d. Compound e. Molecule f. Mixture g. Flocculation h. Gel i. Foam j. Emulsion k. Sol.
-
Use the kinetic theory to explain the following: a. When you take a block of butter out of the fridge, it is quite hard. However, after 15 minutes it is soft enough to spread. b. When you come home...
-
In a particular diffraction grating pattern, the red component (700 nm) in the second-order maximum is deviated at an angle of 20o. (a) How many lines per centimeter does the grating have? (b) If the...
-
The equipment and the accumulated depreciation accounts began the year with balances of $ 5 3 , 5 0 0 and $ 1 9 , 9 0 0 , respectively During the year 2 4 , 0 0 0 of equipment was budgeted for...
-
Use the logical reasoning strategy. Mika and Pazi each think of a number. Mika's number is 7 more than Pazi's number. The sum of the two numbers is 49 . What is Pazi's number?
-
Auditors do not test every document or transaction. How do they gain reasonable assurance through sampling and testing?
-
60 months You want to buy a car and a local bank will lend you $18,500. The loan will be fully amortized over 5 years, and the nominal interest rate would be 4.65%, with interest paid monthly. What...
-
Consider economies in which individuals differ both in their wage and their preferences. Assume we want to compensate them for their wage, while staying neutral with respect to their preferences. In...
-
(a) Find the exact solution to the linear system (b) Solve the system using Gaussian Elimination with 2 digit rounding. (c) Solve the system using Partial Pivoting and 2 digit rounding. (d) Compare...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Prove that the perfect gas temperature scale and the thermodynamic temperature scale based on the Second Law of thermodynamics differ from each other by at most a constant numerical factor.
-
Evaluate (ClS/ClV)]' for (a) A van der Waals gas, (b) A Dieterici gas (Table 1.7). For an isothermal expansion, for which kind of gas (and a perfect gas) will /).5be greatest? Explain your conclusion.
-
Two of the four Maxwell relations were derived in the text, but two were not. Complete their derivation by showing that (S/V)T = (p/T)V (T/P)s = (V/S)p
-
Nichols Inc. manufactures remote controls. Currently the company uses a plant-wide rate for allocating manufacturing overhead. The plant manager is considering switching-over to ABC costing system...
-
a) "When choosing the appropriate group of risk factors to include in a multifactor Arbitrage Pricing Theory (APT) model, we must allow for both unsystematic and systematic sources of risk." Comment...
-
As of December 31, salaries of $9,000 are earned by employees but not yet paid. The company has earned (but not recorded) $950 of interest revenue for the year ended December 31. The interest payment...

Study smarter with the SolutionInn App


