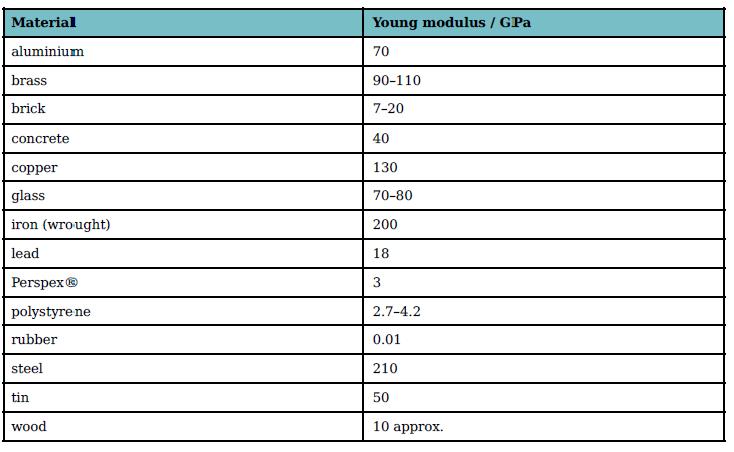
List the metals in Table 7.1 from stiffest to least stiff. Material Young modulus / GPa aluminium
Question:
List the metals in Table 7.1 from stiffest to least stiff.
Transcribed Image Text:
Material Young modulus / GPa aluminium 70 brass 90-110 brick 7-20 concrete 40 copper 130 glass 70-80 iron (wrought) 200 lead 18 Perspex® polystyre ne 2.7-4.2 rubber 0.01 steel 210 tin 50 wood 10 approx.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
List the metals in Table 71 from stiffest to least stiff ...View the full answer

Answered By

Nikka Ella Clavecillas Udaundo
I am a hard-working individual with vast experience in essay, blog, and article writing. I possess skills in network configuration, troubleshooting and managing it, research, content writing(tech, academic, and marketing) and web testing skills. I provide quality work with 100% accuracy, efficiency, and delivery within the stipulated timeline
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Question Posted:
Students also viewed these Sciences questions
-
In Figure a stiff wire bent into a semicircle of radius a = 2.0 cm is rotated at constant angular speed 40 rev/s in a uniform 20mT magnetic field. What are the (a) Frequency and (b) Amplitude of the...
-
List 1 has 13 terms from this chapter, Chapter 1, or Chapter 3; List 2 contains 10 definitions or explanations of terms. Match the definitions with the terms by placing a capital letter from list 1...
-
In theory, most metals should easily corrode in air. Why? A group of metals called the noble metals are relatively difficult to corrode in air. Some noble metals include gold, platinum, and silver....
-
Q4 11 Points 4. Consider the following Current Data for ABC Corp.: Debt Equity Market Value (in millions of $) 16,000 26,000 Cost 7% 14% If ABC moves to and maintains a debt-to-value (D/V) ratio of...
-
New vehicle lift equipment with a value of $82 000 has been purchased by Lockwood Automotive. Starting in 18 months, the company will make payments of $2200 at the beginning of every month over 4...
-
Larned Corporation recorded the following transactions for the just completed month. a. $ 80,000 in raw materials were purchased on account. b. $ 71,000 in raw materials were requisitioned for use in...
-
Early Start Bakery mass-produces bread using three sequential processing departments: Mixing, Baking, and Packaging. The following transactions occurred during February: Requirements: 1. Post each of...
-
Six processes are to be laid out in six areas along a long corridor at Linda Babat Accounting Services. The distance between adjacent work centers is 40 feet. The number of trips between work centers...
-
a) Investment in real estate is one of the booming business in Kenya today and most of companies and wealthy individuals have been venturing into the business. Understanding on the rights of real...
-
Identify the unusual values of x in each histogram in Exercises 68. Data from exercises 6-8: 7. 6. 8. P(x) P(x) P(x) 0.40- 0.40 0.30 0.40 0.30 0.30 0.20 0.10 0.20 0.20 0.10 0.10 O 2 4 6 8 10 12 0 2 4...
-
Which of the non-metals in Table 7.1 is the stiffest? Material Young modulus / GPa aluminium 70 brass 90-110 brick 7-20 concrete 40 copper 130 glass 70-80 iron (wrought) 200 lead 18 Perspex polystyre...
-
A single cell of e.m.f. 1.5 V is connected across a 0.30 resistor. The current in the circuit is 2.5 A. a. Calculate the terminal p.d. and explain why it is not equal to the e.m.f. of the cell. b....
-
A girl stands at A on a platform which is rotating with angular acceleration and at the instant shown has angular velocity .If she walks at constant speed v measured relative to the platform,...
-
Ethane is produced from the hydrogenation of acetylene. Acetylene and hydrogen are fed to a reactor in stoichiometric ratio . The product stream is analyzed and found to contain 20% by mole...
-
Assume that the Incentive Pool in Halifax Hospital only included revenues with respect to the professional component of the oncology services. Would the court have reached a different conclusion?...
-
5. Explain a general approach to determining a limit. 1 6. What is the equation of the normal to y = x-at (1, 0)? Write your answer in the form y = mx + b. x-at X
-
Terry and Maria Redlin have been married for eight years. Terry is an oil rig maintenance supervisor for Holyann Corporation and Maria works as a cashier for Mynards. Terry s birthdate is February 2...
-
You have been asked to present your plan for the proposed health care facility to the Board of Directors. Based on your previous assignments, you are to develop a proposal as a written paper that...
-
A square loop of wire with side 0.60 m carries a current of 9.0 A as shown in the figure. When there is no applied magnetic field, the plane of the loop is horizontal and the non conducting, non...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Draw bond-line structures for all constitutional isomers of the following compound: CH 3 CH 2 CH(CH 3 ) 2
-
In each of the following compounds, identify all carbon atoms that you expect will be deficient in electron density (δ+). If you need help, refer to Section 1.5. a. b. c. Refer section...
-
Atenolol and enalapril are drugs used in the treatment of heart disease. Both of these drugs lower blood pressure (albeit in different ways) and reduce the risk of heart attack. Using the following...
-
For each line, count squares to determine the rise and the run, and calculate the slope. a) b) c) d)
-
Normal retirement benefit: 2% of the average annual salary over the three years prior to that date multiplied by the number of years of service as of that date. The accrued benefit is payable...
-
Under what circumstances would a system administrator lose access to an instance of SQL Server? Give an example.

Study smarter with the SolutionInn App


