a. Draw a diagram of an electrochemical cell consisting of a Cr 3+ /Cr half-cell and a
Question:
a. Draw a diagram of an electrochemical cell consisting of a Cr3+/Cr half-cell and a Cl2/Cl– half-cell.
b. Use the data in Appendix 2 to calculate the cell voltage.
c. Which half-cell is the positive pole?
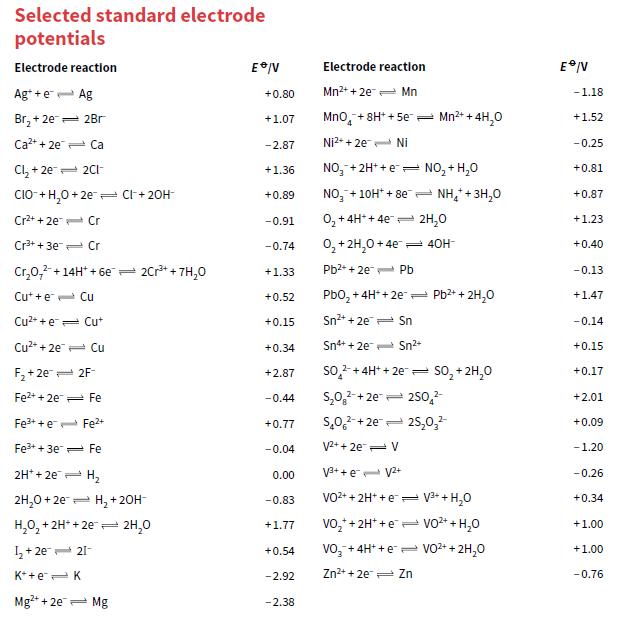
Transcribed Image Text:
Selected standard electrode potentials Electrode reaction Electrode reactlon Ag* +e- Ag Mn2+ + 2e- Mn +0.80 -1.18 Br, + 2e= 2Br +1.07 Mno, + 8H* + 5e- Mn2+ + 4H,0 +1.52 Ca2+ + 2e Ca -2.87 Ni2+ + 2e Ni - 0.25 Cl, + 2e 2CI- NO,-+ 2H+ +e - NO, + H,0 +0.81 +1.36 CIO- + H,0 + 2e Cl+ 20H NO, + 10H* + 8e- NH,+3H,0 +0.89 +0.87 Cr2+ + 2e Cr -0.91 0, + 4H* + 4e= 2H,0 +1.23 Cr3+ + 3e Cr 0, + 2H,0 + 4e 40H- -0.74 +0.40 Cr,0,2-+14H* + 6e- 2Cr+ + 7H,0 Pb2* + 2e Pb +1.33 -0.13 Cut + e- Cu Pbo, +4H* + 2e= Pb2+ + 2H,o +0.52 +1.47 Cu* + e= Cut +0.15 Sn2+ + 2e Sn -0.14 Cu2+ + 2e Cu +0.34 Sn+ + 2e Sn2+ +0.15 F, + 2e 2F so, + 4H* + 2e so, + 2H,0 +2.87 +0.17 Fe2+ + 2e Fe -0.44 S,0,-+ 2e= 250,2 +2.01 Fe3+ + e Fe2+ s,0, +2e= 25,0, +0.77 +0.09 Fe+ + 3e= Fe -0.04 V2+ + 2e- V -1.20 2H* + 2e H, V3+ + e- V2+ 0.00 -0.26 2H,0 + 2e= H, +20H- VO2+ + 2H* +e = V3+ + H,0 +0.34 -0.83 H,0, + 2H* + 2e 2H,0 vo,* + 2H* +e vo2* + H,0 +1.00 +1.77 I, + 2e 21- Vo,-+ 4H* + e e VO2+ + 2H,0 +0.54 +1.00 K* +e=K -2.92 Zn2+ + 2e Zn -0.76 Mg+ + 2e- Mg -2.38
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a The diagram of an electrochemical cell consisting of a Cr3Cr halfcell and a Cl2Cl halfcell ca...View the full answer

Answered By

Mary Boke
I have teached the student upto class 12th as well as my fellow mates.I have a good command in engineering,maths and science.I scored 90+ marks in 10th and 12th in maths.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
a. Draw a diagram of an electrochemical cell consisting of a Mn 2+ /Mn half-cell and a Pb 2+ /Pb half-cell. b. Use the data in Appendix 2 to calculate the cell voltage. c. Which half-cell is the...
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
The data in Appendix D (available at www.uvm.edu/~dhowell/fundamentals8/DataFiles/Add.dat) are actual data on high school students. What is the 75th percentile for GPA in these data? (This is the...
-
On 28 April 2020, Mr Guna, CEO of Econ Engineering Malaysia, proposed to complete an abandoned boiler project that no one had dared to revive. He knew that the project was 60% complete before it was...
-
Let z be a random variable with a standard normal distribution. Find the indicated probability and shade the corresponding area under the standard normal curve. P( 0.45 z 2.73)
-
There is an 82 percent chance the project below can be completed in X weeks or less. What isX? MosT MOST OPTIMISTIC MosT LIKELY PESSIMSTIC ACTIVITY 10 10
-
In Figure \(8.10 a\), how does the magnitude of the downward force exerted by the spring on the ceiling compare with the magnitudes of the downward gravitational forces exerted by Earth on the spring...
-
The following balances were taken from the general ledger of Holland Company on January 1, 2012: During 2012, the company completed the following transactions: a. Purchased inventory for $95,000 on...
-
12. "New competition is not between what companies produce in their factories, but between what they add to their factory output in the form of packaging, services, advertising, customer advice,...
-
Alaskan Railroad is an independent, stand-alone railroad operation not connected to any other rail service in North America. As a result, rail shipments between Alaska and the rest of North America...
-
Show, with the aid of a diagram, how you would measure the E Q value for the half-cell shown by the equation: VO 2+ + 2H + + e V 3+ + H 2 O
-
State the direction of the electron flow in the electrochemical cells represented by the following pairs of half-equations. Use the data in Appendix 2 to help you. a. F 2 + 2e 2F and Mn 2+ + 2e ...
-
What advantages do credit unions have that banks consider unfair?
-
three pages on the working conditions and Workers during the 1800s. What role did union play in organizing workers? Did the Union succeed in organizing workers then? Where did the Union power lie?...
-
The Wakanda Republic is an authoritarian state that exercises so much control and imposes laws without the approval of the people it governs. They support policies that benefit unitarist employers,...
-
What was the French Revolution and which condition do you personally think was the most important in allowing it to come about? In your opinion, what was the most shocking example of violence during...
-
Juan owns a restaurant and his electricity bill doubled. To offset this cost Juan now has to raise the prices on his menu. This is an example of what factor of supply? Explain
-
Iron ore is used to make steel. If a new source of iron ore was discovered in the United States, how might that affect the supply of cars?
-
Photons of energy E = 4.000 keV undergo Compton scattering. What is the largest possible change in photon energy, measured as a fraction of the incident photon's energy (E E) / E?
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
In an adiabatic compression of one mol of an ideal gas with C V ,m = 5/2 R, the temperature rises from 278 K to 450. K. Calculate q, w, H, and U.
-
Can the following compound be prepared via a Williamson ether synthesis? Explain your answer.
-
Calculate H o R and U o R for the oxidation of benzene (g). Also calculate -U R.
-
Factor the following polynomial. 8m(n-3)-(n-3)
-
2 For the function f(x) = x+5x-14 solve the following. f(x) 0
-
Find the product in the form C. x. y. The coefficient C = and the exponents are a = and b = Note: You can earn partial credit on this problem. (-10xy)(-6xy)

Study smarter with the SolutionInn App


