Look at the two infra-red spectra below: a. Which one of the infra-red spectra is that of
Question:
Look at the two infra-red spectra below:
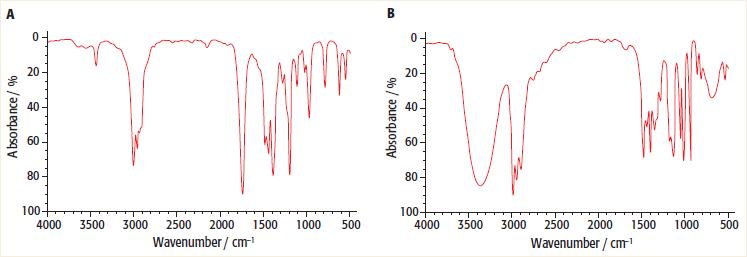
a. Which one of the infra-red spectra is that of butanone and which one is of butan-2-ol?
b. Explain your reasoning in part a.
Transcribed Image Text:
В 20 20- 40 40- 60 60 80 08 100 4000 100 3500 3000 2500 2000 1500 1000 500 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber / cm- Wavenumber / cm A bsorbance/ %
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a Image A is for Butanone image B is for Butan 2ol b In image A there ...View the full answer

Answered By

Manasa D K
I am a MSc graduate, cleared lectureship eligibility test. I has a 5 years of experience in teaching chemistry for 11th and 12th, BSc and MSc graduates. My skills include, proficiency in a wide range of teaching methods and techniques. Extensive hands on experience in the practice of chemistry and solid grasp of its theories. Comprehensive knowledge and understanding of the subject to creat, execute and analyse student assessments.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Look at the two histograms, created from 2009 real estate data taken from the Ventura County Star, and decide whether you think the standard deviation of home prices in Agoura, California (A), was...
-
One of the basic financial principles is that the value of any asset (whether it be a stock, a bond, or a firm as a whole) is the present value of that asset's future cash flows. As you learned in...
-
Which one of the following press types is usually associated with the highest production rates in sheet-metal-stamping operations: (a) adjustable bed, (b) open-back inclinable, (c) press brake, (d)...
-
Find the laurents series f(z)= 1/(z-1)(z-2) (i) |z| <1 (ii) 1 2
-
The following is based on information from The Wolf in the Southwest: The Making of an Endangered Species by David E. Brown (University of Arizona Press). Before 1918, the proportion of female wolves...
-
Prepare the monthly income statement, monthly statement of owners equity, and the balance sheet at August 31, 2014, for Krishna Cleaning Company from the data provided in the adjusted trial balance...
-
Refer to the Arctic Springs Filtration Department Data Set. At Arctic Springs, water is added at the beginning of the filtration process. Conversion costs are incurred evenly throughout the process,...
-
For financial reporting, Clinton Poultry Farms has used the declining-balance method of depreciation for conveyor equipment acquired at the beginning of 2008 for $2,560,000. Its useful life was...
-
1. Electric potential is given by V=6x-8xy-8y+6yz - 4z Then magnitude of electric force acting on 2C point charge placed on origin will be :- (1) 2N (2) 6N (3) 8N (3) 8N (4) 20 N 2. Figure shows...
-
John and Sandy Ferguson got married eight years ago and have a seven-year old daughter Samantha. In 2014, John worked as a computer technician at a local university earning a salary of $52,000, and...
-
An alcohol has the molecular formula C 3 H 8 O. When warmed with an alkaline solution of iodine it forms a yellow precipitate. a. Name the yellow precipitate. b. Draw the displayed formula of the...
-
Use data from the table below of characteristic infra-red absorptions in organic molecules to answer the following question. One of the three spectra labelled A to c below is produced when ethanal is...
-
Consider a large country applying a tariff t to imports of a good like that represented in Figure 8-9. How does the size of the terms-of-trade gain compare with the size of the deadweight loss when...
-
1. Calculate f for a nachannel MOSFET whose T Cas 6.1 f F, C=1.7fF capacitances are given given as Cgs gd Assume operation current at 100 A, and that Kn= 160 A/V, W = 10m, L= 1m.
-
In 2023 an employee drives a delivery van owned by the employer. The van does not meet the definition of an "automobile". The employee's personal use is 7,000 kilometres. Following are some rates you...
-
Answer: Bad debt and doubtful debt are terms used in accounting to classify debts that a company is unlikely to collect from its customers. Here's how they differ: Bad Debts: This alludes to the...
-
Heads-Up Company sold 7,000 scooter helmets at $96.00 each this fiscal year. Unit variable costs were $60.00 (includes direct material, direct labor, variable manufacturing overhead, and variable...
-
WT has owned 75% of BD for a number of years. In the year to 31 December 20X3 WT had revenue of $350,000 and BD had revenue of $125,000. Included in WT's revenue is goods sold to BD for $25,000 that...
-
Why can a neutron within a nucleus be stable, while an isolated neutron is unstable? What determines whether a neutron within a nucleus is stable?
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
A 1.50 mole sample of an ideal gas at 28.5C expands isothermally from an initial volume of 22.5 dm 3 to a final volume of 75.5 dm 3 . Calculate w for this process a. For expansion against a constant...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Consider the reaction TiO 2 (s) + 2 C(graphite) + 2 Cl 2 (g) 2 CO(g) + TiCl 4 (l) for which ÎH o R ,298 K = 80. kJ mol 1 . Given the following data at 25°C, Assume that the heat capacities...
-
An accounting department is preparing the year-end warranty accrual. They ask the warranty department to provide past data on the dates, part numbers, and the related repair costs (including both...
-
Donna Company began operations on June 1. The following transactions took place in June: a. Purchases of merchandise on account were $750,000. b. The cost of freight to receive the inventory was...
-
According to ASU 2020-07, how must contributed nonfinancial assets such as land, buildings, materials, and the use of facilities be presented in a Statement of Activities?

Study smarter with the SolutionInn App


