Calculate the standard heats and Gibbs energies of reaction at 25C for the following reactions: a. N(g)
Question:
Calculate the standard heats and Gibbs energies of reaction at 25°C for the following reactions:
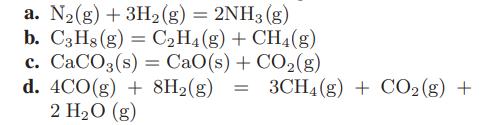
Transcribed Image Text:
a. N₂(g) + 3H₂(g) = 2NH3 (g) b. C3H8 (g) = C₂H4 (g) + CH4(g) c. CaCO3(s) = CaO(s) + CO₂(g) d. 4CO(g) + 8H2(g) = 3CH4(g) + CO₂(g) + 2 H₂O (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the standard heats AH and Gibbs energies AG of reaction at 25C for the given reactions ...View the full answer

Answered By

Caroline Kinuthia
Taking care of the smaller details in life has a larger impact in our general well being, and that is what i believe in. My name is Carol. Writing is my passion. To me, doing a task is one thing, and delivering results from the task is another thing. I am a perfectionist who always take things seriously and deliver to the best of my knowledge.
4.90+
1933+ Reviews
4273+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Sn4+(aq) + 2Hg(l Sn2+(aq) + Hg2 2+(aq) The equation is not balanced.
-
Calculate the standard free-energy change at 25C for the following reaction. 4Al(s) + 3O2(g) + 12H + (aq) 4Al3 + (aq) + 6H2O(l) Use standard electrode potentials.
-
Mookie The Beagle Concierge Trial Balance As of January 31,2023 is given 1001 Checking 1010 Money Market 1100 Accounts Receivable (A/R) 1210 Prepaid Expenses:Supplies 1220 Prepaid Expenses:Insurance...
-
Alex is a wage earner with a regular income. He has unsecured debts of $42,000 and secured debts owing to Betty, Connie, David, and Eunice totaling $120,000. Eunices debt is secured only by a...
-
The present value model predicts the following relationship between the two series \[ p_{t}=\beta_{0}+\beta_{1} d_{t}+u_{t} \] where \(p_{t}\) is the natural logarithm of real price of equities,...
-
Show that the real component of the harmonic response of a viscously damped single-degreeof-freedom system (from \(X\) in Eq. (3.54)) attains a maximum at...
-
Jack Jones, the materials manager at Precision Enterprises, is beginning to look for ways to reduce inventories. A recent accounting statement shows the following inventory investment by category raw...
-
If your organization was given $10,000 for digital marketing for a quarter (3 months), how would you spend it?
-
PLEASE PREPARE THE COMPARATIVE FINANCIAL STATEMENTS OF THE FOLLOWING: Statement of Changes in Shareholders Equity Statement of Cash Flows. ALL INFORMATION ARE GIVEN ABOVE Fall Bake Shop Corporation...
-
Calculate the minimum work required to separate air (79 mole % nitrogen) into pure oxygen and nitrogen assuming an isothermal, steady flow process at 300 K. The inlet air pressure is 10 bar and each...
-
a. Given experimental data either for the excess Gibbs energy, G ex , or for species activity coefficients from which G ex can be computed, it is sometimes difficult to decide whether to fit the data...
-
Mr. and Mrs. Kitchens purchased their first home in Ohio for $135,000 on October 1, 2016. Because Mr. Kitchens employer transferred him to Utah, they sold the house for $160,000 on January 10, 2017....
-
George Kritopoulos, 50, Salem, Massachusetts, a real estate developer was sentenced to four years in prison today in connection with a decade-long mortgage fraud scheme involving at least two dozen...
-
One example provided by our guest lecturer, Eliana Hruby, is the use of RPA to generate supplier statements to send to the University's suppliers. (a) Explain how this process change enables greater...
-
In the complex numbers, where i = -1, what complex number x is a solution to the equation x(2 + 3i) = 1?
-
Compute ending work in process inventory for a manufacturer using the following Raw materials purchased Direct materials used Direct labor used Factory overhead Work in process inventory, beginning...
-
Complete the followin Common Size Balance Sheet: Amount Percent Cash $ 600 (c)_____________________________ Accounts Receivable 1,400 (d)_____________________________ Total Aseets $ 2,000...
-
How does the regulation of insurance companies compare with that of depository institutions?
-
Sportique Boutique reported the following financial data for 2012 and 2011. Instructions(a) Calculate the current ratio for Sportique Boutique for 2012 and 2011.(b) Suppose that at the end of 2012,...
-
Use MATLAB to calculate a. b. c. d. Check your answers with a calculator. el-2.1 + 3.47 log(14) + V287
-
Use MATLAB to calculate a. 6 tan -1 (12.5) + 4 b. 5 tan [3 sin -1 (13/5)] c. 5 ln(7) d. 5 log(7) Check your answers with a calculator.
-
The Richter scale is a measure of the intensity of an earthquake. The energy E (in joules) released by the quake is related to the magnitude M on the Richter scale as follows. E = 10 4.4 10 1.5M How...
-
1. The marital deduction: a. What is the marital deduction? b. How is the marital deduction "unlimited"? c. What requirements must be met in order to obtain the marital deduction? d. Why does the use...
-
Fiori Corporation's relevant range of activity is 3,100 units to 6,500 units. When it produces and sells 4,800 units, its average costs per unit are as follows: Average Cost per Unit Direct materials...
-
In this group project, you are required to conduct a legal analysis and develop a "legal argument" WHICH is merely a legal analysis. The premises of a legal argument consist of facts, laws, your...

Study smarter with the SolutionInn App


