Derive the analogues of the Gibbs-Duhem equations (Eqs. 8.2-8 and 8.2-9) for the constraints of a.Constant temperature
Question:
Derive the analogues of the Gibbs-Duhem equations (Eqs. 8.2-8 and 8.2-9) for the constraints of

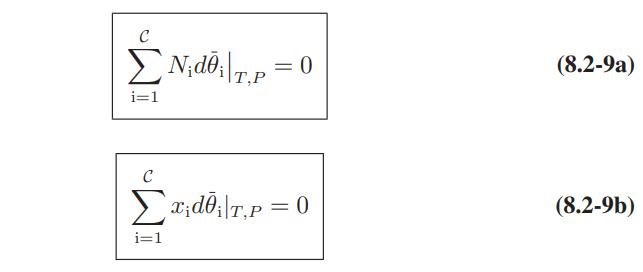
a.Constant temperature and volume
b. Constant internal energy and volume
c. Constant entropy and volume
Transcribed Image Text:
-N от P,Ni C v (OP) ₁ dP+ ► N; d0; = 0 T,Ni i=1 dT - N N 20 ( ) ат - (р) dT ӘT P,Ni T,Ni C dP+Ex; d0; = 0 i=1 (8.2-8a) (8.2-8b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
We have the following properties for a certain mixture for mixing at constant temperature and pressure: where S i , the pure-component molar entropy of component i, is given by Here S i , U i , and V...
-
For a certain company, the cost function for producing x items is C(x)=30x+100 and the revenue function for selling x items is R(x)=0.5(x70)^2+2,450 . The maximum capacity of the company is 100...
-
The shareholders of Endicott Johnson who had dissented from a proposed merger of Endicott with McDonough Corporation brought a proceeding to fix the fair value of their stock. At issue was the proper...
-
The number of degrees of freedom for a system consisting of the gases \(\mathrm{CO}, \mathrm{CO}_{2}, \mathrm{H}_{2}\), \(\mathrm{H}_{2} \mathrm{O}\) and \(\mathrm{CH}_{4}\) in chemical equilibrium...
-
When a transducer is used in conjunction with another device to measure vibration, it is called a a. vibration sensor b. vibration pickup c. vibration actuator
-
Richard, age 40, is the owner of Auto Repair, Inc. In addition to Richard, the company has five employees. Richard wants to establish a retirement plan for his employees. He is considering two plans:...
-
What are secondary publics for a candle company? Examples of secondary publics .
-
Happyville Hospital and Sunnyville Hospital are located across the street from each other, and the respective hospital boards have voted to merge the facilities into the Limestone Health Care System....
-
Estimate the vapor pressures of the refrigerants 1,1,1,2-tetrafluoroethane, dichlorodifluoromethane and 2-chloro-1,1,1,2-tetrafluoroethane over the temperature range of 40 to +85C.
-
Explain the nonmonotonic behavior of the fugacity coefficent along the T = 1.50 isotherm in Fig. 7.4-1 using the van der Waals equation of state. Fig. 7.4-1 Fugacity coefficient, 1.5 1.4 1.3 1.2 1.1...
-
During January 2014, Hexagon Company purchased 12,000 of the 200,000 outstanding common shares of Smiley Corporation at $ 30 per share. This block of shares was purchased as a long- term investment....
-
Sheridan Mining Company purchased land on February 1, 2025, at a cost of $975,900. It estimated that a total of 57,600 tons of mineral was available for mining. After it has removed all the natural...
-
Cullumber Company completes and transfers out 15,360 units and has 2,560 units of ending work in process that are 25% complete as to conversion costs. Materials are entered at the beginning of the...
-
a. Journalize the entries to record (1) The declaration of the dividend, capitalizing an amount equal to market value, and (2) The issuance of the stock certificates. b. Determine the following...
-
Record the following transactions on the books of Ivanhoe Co. (Credit account titles are automatically indented when amount is entered Do not indent manually.) a. On July 1, Ivanhoe Co. sold...
-
?Journalize Loondance ' s July transactions. ( You do not need to record the cost of goods sold or inventory entry or ?entries. ) ( Record debits ?first, then credits. Exclude explanations from any...
-
What is the contribution to the asset base of the following items under the Basel III requirements? a. $ 10 million cash reserves. b. $ 50 million 91-day U. S. Treasury bills. c. $ 25 million cash...
-
2. In the circuit given in Figure 2, i,(t) = 5.67cos(5t)A and v (t) = 70.71 cos(5t 60) V a) Find the equivalent load impedance. State whether the load is inductive or capacitive. b) Calculate the...
-
Determine the elastic modulus and the yield strength for the material having the stressstrain curve shown in Figure P5.11. Use the 0.2% offset method. Figure P5.11 1600 1400 1200 1000 800 600 400...
-
A 1-ft-long rod is made from the material of Problem P5.11. By what amount must the rod be stretched from its original length for it to begin yielding? Problem P5.11 Determine the elastic modulus and...
-
The steel bolt and anchor assembly is used to reinforce the roof of a passageway in an underground coal mine (Figure P5.13). In installation, the bolt is tensioned to 5000 lb. Calculate the stress,...
-
Below is a summary of a CPM for southwest airlines, Alaska and American Airlines. Please provide a summary. the main firm with the weigh score is southwest airlines. What does this mean for southwest...
-
1. (25 marks) The financial year end for a large machine parts manufacturer will end in a week's time. The manager, Rebecca who is in charge of 10 salespersons is overwhelmed with work as is usual...
-
INFORMATION As a management accountant, you have recently been recruited to be part of the strategic management accounting team of GHK Accountants. GHK Accountants has been contracted to help...

Study smarter with the SolutionInn App


