Derive the equation that replaces Eq. 13.5-16 if the thermodynamic equilibrium constant (rather than the apparent equilibrium
Question:
Derive the equation that replaces Eq. 13.5-16 if the thermodynamic equilibrium constant (rather than the apparent equilibrium constant) is used and electrolyte solution nonideality is included.
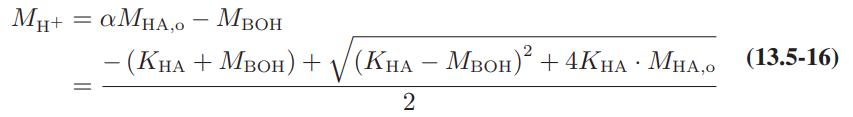
Transcribed Image Text:
MH+ = aMHA,0 - MBOH -(KHA+MBOH) + (KHA - MBOH)2 + 4KHA MHAO (13.5-16) 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To derive the equation that replaces Eq 13516 when using the thermodynamic equilibrium constant and ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Derive the equation that replaces Eq. 13.5-11 if the thermodynamic equilibrium constant (rather than the apparent equilibrium constant) is used and electrolyte solution nonideality is included. pH =...
-
Derive the equations that replace Eqs. 13.5-12a and b if the thermodynamic equilibrium constant (rather than the apparent equilibrium constant) is used and electrolyte solution nonideality is...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Conduct a search of position announcements by companies that are recruiting expatriates for foreign subsidiaries. Based on your findings, what seem to be some of the shared features contained in...
-
Homestead Pottery, Inc., is divided into two operating divisions: pottery and retail. The company allocates power and human resources department costs to each operating division. Power costs are...
-
The budget director of Feathered Friends Inc., with the assistance of the controller, treasurer, production manager, and sales manager, has gathered the following data for use in developing the...
-
Acne Medication and Gastrointestinal Injury Since 1999, Kamie Kendall had experienced ongoing abdominal pain, and she was eventually diagnosed with ulcerative colitis and irritable bowel syndrome...
-
Gross Profit Method Astaire Company uses the gross profit method to estimate inventory for monthly reporting purposes. Presented below is information for the month of May. Inventory, May...
-
x-5 and justify your answer. Respond in the space 3x-1 f(x)= Determine the range of provided below.
-
Derive the equation that replaces Eq. 13.5-16 for the case of a weak base and a strong acid. MH+ = aMHA,0 MBOH -(KHA +MBOH)+ (KHA - MBOH) +4KHA MHA,O (13.5-16) 2 .
-
Redo Illustration 13.5-2 including the ionization of water and including the effect of solution nonidealities. Illustration 13.5-2 A common analytical procedure is titration, in which a solution of a...
-
A city enters into an 11 month lease with an option to renew the lease for additional 11 months up to 5 times. In your opinion does the lease qualify for the one-year exception and thereby be...
-
You work for a U.S. firm, and your boss has asked you to estimate the cost of capital for countries using the euro. You know that S = $1.1625/; and F1 = $1.1617/;. Suppose the dollar WACC for your...
-
IZAX, Co. had the following items on its balance sheet at the beginning of the year:Its net income this year is $19,600 and it pays dividends of $5100. If its assets grew at its internal growth rate,...
-
Etemadi Amalgamated, a U.S. manufacturing firm, is considering a new project in Portugal. You are employed in Etemadis corporate finance department and are responsible for deciding whether to...
-
Kurta Company Ltd. has a budget of $10 million for new projects. The projects are independent and have the following costs and profitability indexes associated with them: a. Make your selection under...
-
Alfa trading operates a quite successful chain of milk products and tea houses across the South Caucasus, Alfa plans to expand in Ukraine and Moldova, so it needs to raise some funds. The Companys...
-
Use the information in Exercise 14-14 to prepare the journal entries for Eagle to record the loan on January 1, 2013, and the four payments from December 31, 2013, through December 31, 2016. In...
-
What is the expected payoff of an investment that yields $5,000 with a probability of 0.15 and $500 with a probability of 0.85? Select one: O a. $325 O b. $5,500 O c. $2,750 O d. $1,175
-
Draw the shear and moment diagrams for the beam. 8 kN 15 kN/m 20 kN m B -2 m-f1 m2 m- 3 m-
-
Draw the shear and moment diagrams for the beam. 400 lb/ft 900 lb ft B 4 ft 2 ft- 3 ft-
-
Draw the shear and moment diagrams for the beam. The supports at A and B are a thrust and journal bearing, respectively. 200 N/m B 600 N- m 300 N m 6 m -
-
Suppose the U.S. Treasury auctions $10 billion of 90-day bills, and the competitive bids are $6 billion at 1.670 %, $3 billion at 1.673%, and $2 billion at 1.675%. Noncompetitive bids are $1 billion...
-
Upon reviewing a business rule and ER. diagram related to E-beats_Africa, you observe data redundancy. Conceptualise and describe the data redundancy you observe. Also, illustrate the data redundancy...
-
The Frackville Power Plant combusts methane to produce electricity. The plant burns 37,500,000 cubic yards of methane each year. The plant produces 61,626,000 kWh of electricity. Calculate the...

Study smarter with the SolutionInn App


