Evaluate the difference for the ideal and van der Waals gases, and for a gas that obeys
Question:
Evaluate the difference
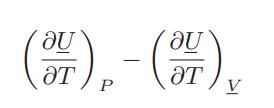
for the ideal and van der Waals gases, and for a gas that obeys the virial equation of state.
Transcribed Image Text:
(3²) - (0)₁ P V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The ideal gas equation is PV nRT where P is the pressure V is the volume n is the number of moles of gas R is the ideal gas constant T is the temperat...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO 2 (g) when it is confined at 298 K to a volume of (a) 100.0 L; (b) 50.0 L; (c) 20.0...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
When they hear the name Credit Suisse, my grandchildren will likely have to google (or whichever search engine exists then) these two words. For most of my contemporaries here in Switzerland, Credit...
-
Universal Video sells $40 million worth of video recording equipment in the United States. The total sales of such equipment in the United States is $100 million. One-half of Universals sales is to...
-
Current assets for two different companies at calendar year-end 2009 are listed here. One is a manufacturer, Nordic Skis Mfg., and the other, Fresh Foods, is a grocery distribution company. (1)...
-
Which item(s) appears as a reconciling item(s) to the book balance in a bank reconciliation? a. Outstanding checks b. Deposits in transit C. Both a and b d. None of the above
-
The balance sheet for Throwing Copper, Inc., is shown here in market value terms. There are 23,000 shares of stock outstanding. The company has declared a dividend of $1.35 per share. The stock goes...
-
How does the dynamic interplay between transformational and transactional leadership styles influence organizational adaptability in complex business environments ?
-
John and Barbara were first cousins living in Washington. Under Washington law, they were not permitted to marry. They traveled to Nashville, Tennessee, one weekend and married in a courthouse...
-
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in...
-
For steam at 500 C and 10 MPa, using the Mollier diagram, a. Compute the Joule-Thomson coefficient = (T/P) H . b. Compute the coefficient S = (T/P) S . c. Relate the ratio ( H / S ) T /(H/S) P to ...
-
Suppose the number (in millions) of bacteria present in a culture at time \(t\) is given by the formula \[N(t)=2 t^{2}-200 t+1,000\] Use this formula in Problems 57-60. Find the instantaneous rate of...
-
Jorge and Anita, married taxpayers, earn $151,600 in taxable income and $40,400 in interest from an investment in City of Heflin bonds. Using the U.S. tax rate schedule for married filing jointly,...
-
17. What is meant when a persons mass is stated as 73.6 kg? That A. the mass is closer to 73.6 than it is to 73.7 or 73.5 B. the mass is closer to 73.6 than to any other value C. the mass is between...
-
Find the net electric flux through the closed spherical surface in a uniform electric field shown in Figure a a 4R E 2R E TRE R
-
Consider a project that requires spending of $15,000 immediately and generates $6,000 per year for the next 4 years (assume each $6,000 comes at the end of each year).What is the NPV of this project?...
-
21. Which of the following measurements is a semi-quantitative one? A. a blood pressure of 120/80 mmHg B. a blood glucose level of + + + C. a state of anxiety measure of calm D. the patients name is...
-
Karen Martinis was responsible for opening mail, processing vendor claims, and authorizing payments. She was involved in a scheme in which she either double-paid vendor invoices, paid the wrong...
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
The concept of thermal resistance described in Problem 8 can be used to find the temperature distribution in the at square plate shown in Figure P9(a). Figure P9 The plates edges are insulated so...
-
Compute the double integral Note that the region of integration lies to the right of the line y = x. Use this fact and a MATLAB relational operator to eliminate values for which y > x. A = 2 (x +...
-
Compute the triple integral A = xe dxdy dz Jo Ji 0, 2.
-
Coronado's CVP income statement included sales of 6000 units, unit selling price of $100, unit variable cost of $60, and fixed expenses of $160000. Contribution margin is
-
How do sociological concepts such as socialization, social control, and deviance shed light on the construction and enforcement of social norms, moral codes, and legal systems within diverse cultural...
-
If GDP during that same time period is equal to $120 million, what unplanned changes in inventories occurred? Consumption is $70 million, planned investment spending is $25 million, government...

Study smarter with the SolutionInn App


