L-cystine C 6 H 12 N 2 O 4 S 2 has four ionization states with the
Question:
L-cystine C6H12N2O4S2 has four ionization states with the following values for its ionization constants:
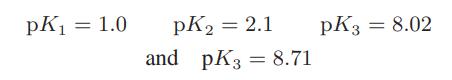
Determine the charge on L-cystine as a function of pH.
Transcribed Image Text:
pK₁ = 1.0 pK₂ = 2.1 and pK3 = 8.71 pK3 = 8.02
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
The charge on Lcystine as a function of pH is determined by the ionization states of its functional ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Oxaloacetic acid C 4 H 4 O 5 has the following values for its ionization constants: Determine the charge on oxaloacetic acid as a function of pH. pK = 2.55 pK = 4.37 and and pK3 pK3 13.03 =
-
Oxalic acid C 2 H 2 O 4 is a dibasic acid with the following values for its ionization constants: Determine the charge on oxalic acid as a function of pH. pK = 1.2 and pK = 4.2
-
A firm had the following values for the four debt ratios discussed in the chapter: Liabilities to Assets Ratio: less than 1.0 Liabilities to Shareholders Equity Ratio: equal to 1.0 Long-Term Debt to...
-
Determine one positive and one negative coterminal angle for each angle given. 173
-
Explain why it is better to allocate budgeted support service costs rather than actual support service costs.
-
Consider steam at the following state near the saturation line: (p1, T1) = (1.31 MPa, 290C). Calculate and compare, for an ideal gas (Table A.4) and the Steam Tables (or the EES software), (a) the...
-
For an ideal solution, the value of the activity coefficient is (a) 0 (b) 1 (c) \( <1\) (d) \(>1\).
-
Inglis City had a beginning cash balance in its enterprise fund of $895,635. During 2014, the following transactions occurred: 1. Interest received on investments totaled $42,400. 2. The city...
-
5. A thin insulating rod with uniformly spread positive charge Q is bent into a circle of radius R. The center of the circle is z = 0. a. Without calculation, roughly plot the magnitude of the E...
-
Creatine C 4 H 9 N 3 O 2 has been used by body builders and athletes to increase muscle mass and strength. Its chemical structure (not showing all of its carbon atoms and attached hydrogens) is The...
-
a. Prove that the isoelectric point for an amino acid with two ionization sites is b. Develop an equation for the calculation of the isoelectric point of an amino acid or protein with three...
-
Lisa Kim, the new controller of LafrenieÌre Company, has reviewed the expected useful lives and residual values of selected depreciable assets at December 31, 2014. (Depreciation for 2014 has...
-
A gas station owner finds that for every penny increase in the price of gasoline, she sells 1657 fewer gallons per week. Using x to represent the number of penny changes in price, create an equation...
-
My chosen project is Beat the Clock: Time Management Training Can Improve Productivity and Morale by Helping Employees Balance Work and Family byKathryn Tyler Address the following items, which will...
-
For this assignment, you will be demonstrating that you know (1) how to read the rhetorical situation of applying for a position, and (2) how to write to that situation by presenting yourself...
-
Sterling purchased a 2 0 year annuity. Sterling paid $ 7 5 0 0 0 in 1 year, and each year for the next 2 0 years sterling will recieve $ 1 0 0 0 0 . How much of of each annuity payment does sterling...
-
Write the expression as a single logarithm. Check 1 7 logx- logy +6 log w 2 log (D
-
You have requested and received the following information from your audit client: Number of people employed at the beginning of each month throughout the current and prior year, and their positions...
-
The process of collaborative goal setting by a manager and subordinate, the extent to which goals are accomplished is a major factor in evaluating and rewarding the subordinate's performance. It is...
-
If a tension force T = 215 lb is required to pull the 200-lb force around the collar bushing, determine the coefficient of static friction at the contacting surface. The belt does not slip on the...
-
The vehicle has a weight of 2600 lb and center of gravity at G. Determine the horizontal force P that must be applied to overcome the rolling resistance of the wheels. The coefficient of rolling...
-
The tractor has a weight of 16,000 lb and the coefficient of rolling resistance is a = 2 in. Determine the force P needed to overcome rolling resistance at all four wheels and push it forward. P 2...
-
Swifty Inc. had beginning inventory of $23,000 at cost and $32,000 at retail. Net purchases were $156,302 at cost and $206,000 at retail. Net markups were $11,300, net markdowns were $7,000, and...
-
Prepare journal entries to record the following transactions for a retail store. The company uses a perpetual inventory system and the gross method. April 2 Purchased $6,600 of merchandise from Lyon...
-
Current Year 1 Year Ago 2 Years Ago $ 35,677 64,366 85,055 $ 31,780 91,197 111,223 Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise...

Study smarter with the SolutionInn App


