The amino acid dl-alanine HCOO-CNH 2 H-CH 3 exists in the following four forms in solution Determine
Question:
The amino acid dl-alanine HCOO-CNH2H-CH3 exists in the following four forms in solution
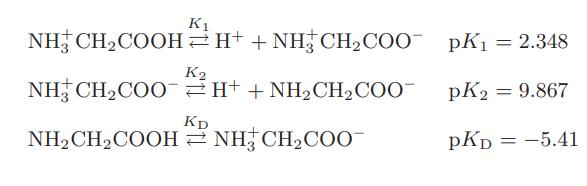
Determine the fraction of dl-alanine in each of these forms at
a. pH = 3
b. pH = 10
Transcribed Image Text:
K₁ NH CH₂COOH H+ + NHCH₂COO- NH CH,COO` =H*+NH,CH,COO KD NH₂ CH₂COOH NH3 CH₂COO- K2 pK₁ = 2.348 pK2 = 9.867 PKD = -5.41
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a At pH 3 the dlalanine will be mostly in the zwitterion form with a small amount in the cationic fo...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The OH group on the side chain of serine is not deprotonated at a pH of 12. However, the OH group on the side chain of tyrosine is deprotonated at a pH of 12. This can be verified by inspecting the...
-
Chi and Daniella are your clients and you are assessing their current financial situation and how it impacts their insurance needs. Chi and Daniella are married and they have a mortgage on their home...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Given that log 10 (x 2 y) = 4 and log 10 (x 4 /y 3 ) = 18, find the value of log 10 x and of log 10 y.
-
Biotechtron, Inc., has two research laboratories in the Midwest, one in Tulsa, Oklahoma, and one in Ames, Iowa. The owner of Biotechtron centralized the legal services function in the Tulsa office,...
-
A 12.0-kg object hangs in equilibrium from a string with a total length of L = 5.00 m and a linear mass density of μ = 0.001 00 kg/m. The string is wrapped around two light, frictionless pulleys...
-
"If the two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other." This is known as the (a) First law of thermodynamics (b) Second law of...
-
Lebo Hardware reported cost of goods sold as follows. Lebo made two errors: (1) 2010 ending inventory was overstated $3,000, and (2) 2011 ending inventory was understated $6,000.InstructionsCompute...
-
A sled is pulled at a constant velocity across a horizontal slippery surface. If a force of 230.0 N is being applied to the sled rope at an angle of 48.0 degrees to the ground, what is the magnitude...
-
Redo Illustration 13.5-2 including the ionization of water and including the effect of solution nonidealities. Illustration 13.5-2 A common analytical procedure is titration, in which a solution of a...
-
Ammonia, air and water is to be converted to a nitric acid solution. Ammonia is available as a liquid at 25C and its saturation pressure, air and water (as a liquid) are available at 1.013 bar and...
-
Find the velocity, acceleration, and speed of a particle with position vector r(t) = t 2 , e t , te t ).
-
During August of the prior year, Julio purchased an apartment building that he used as a rental property. The basis was $ 1 , 4 0 0 , 0 0 0 . Calculate the maximum depreciation expense during the...
-
How do ethical considerations in leadership influence corporate social responsibility, and what frameworks can leaders use to ensure that business decisions align with broader societal values ?
-
What are exempt payments from FICA? What are some examples of exempt payments that you are aware of ? Discuss and provide examples of at least 2 different types of companies that do not withhold...
-
In the context of strategic decision-making, how can leaders incorporate data-driven insights while still allowing for intuition and creativity to play a role in the process ?
-
S owns 1 0 preferred shares of X Corporation with a par value of P 1 0 0 , 0 0 0 per share at 5 % guaranteed cumulative dividends. If after 4 years the corporation decided to declare the regular...
-
What are managements objectives related to payroll?
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
Draw the shear and moment diagrams for the beam. 6 kN/m 3 kN/m 3 m
-
Draw the shear and moment diagrams for the beam. 4 kN/m 2 kN/m B 3 m- 1.5 m
-
Draw the shear and moment diagrams for the beam. 3 kN 6 kN/m B 1.5 m 1.5 m
-
Bogut Company estimates that annual manufacturing overhead costs will be $1,500,000. Estimated annual operating activity bases are direct labor cost $300,000, direct labor hours 15,000, and machine...
-
Performance Products Corporation makes two products, titanium Rims and Posts. Data regarding the two products follow: Direct Labor- Hours per unit Rims Posts 0.70 0.70 Annual Production 26,000 units...
-
How well does this document supports users in taking action or making decisions, as well as how well the document accomplishes inferred purposes of whoever created the document? How this document...

Study smarter with the SolutionInn App


