The following data are available for the mean ionic activity coefficients of these salts in water at
Question:
The following data are available for the mean ionic activity coefficients of these salts in water at 25°C.
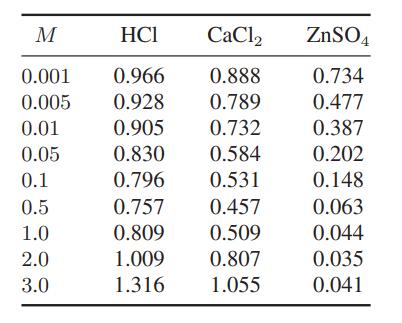
a. Fit these data as best you can using the equations in this chapter for the mean ionic activity coefficient.
b. Determine the activity coefficient of water in each of these solutions.
Transcribed Image Text:
M HCI 0.001 0.966 0.005 0.928 0.01 0.905 0.05 0.830 0.1 0.796 0.5 0.757 1.0 0.809 2.0 1.009 3.0 1.316 CaCl2 0.888 0.789 0.732 0.584 0.531 0.457 0.509 0.807 1.055 ZnSO4 0.734 0.477 0.387 0.202 0.148 0.063 0.044 0.035 0.041
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To fit the data for the mean ionic activity coefficients of the salts in water at 25Cwe can use the ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The following data are available for three companies at the end of their fiscal years: Required Determine the amounts indicated by question marks. Company A $ 600,000 Finished goods, January 1 Cost...
-
(a) Briefly explain what an embedded system is and why we need embedded system based solutions. (b) With the aid of simple functional block diagram, briefly explain how embedded systems work. (c)...
-
The following data are available for Haul-It-Away Truckers: 1. Compute the predetermined overhead rate for each of the two years, if based on (a) Direct labor hours, (b) Number of moving jobs, and...
-
Clark had brain surgery. Insurance will not pay for the surgery until the deductible of $1,000 is hit. Then Clark's coinsurance of 80% / 20% kicks in. Clark has an out-of-pocket maximum of $7,500....
-
Walesco Corporation has decided to discontinue an entire component of its business effective November 1, 2008. It hopes to sell the assets involved and convert the physical plant to other uses within...
-
In designing a relational database, why might we choose a non-BCNF design?
-
In 1988, the Upper Deck Company was a company with an idea for a better baseball card: one that had a hologram on it. By the 1990s, the firm was a major corporation worth at least a quarter of a...
-
Sunshine House received a contract this year as a supplier of Girl Scout cookies. Sunshine currently has five production lines, each of which will be dedicated to a particular kind of ookie. The...
-
Pioneer Inc. wants to invest $557,302 today. The expected returns in years 1, 2, and 3 are $247,615, $180,383, and $335,481, respectively. If the rate of return on investment must be at least 14%,...
-
A thermodynamic property of a mixture is given by a. Develop expressions for the partial molar properties 1 , 2 , and 3 as a function of the pure component molar properties, the mole fractions,...
-
A gas stream at 310 K and 14 bar is to be compressed to 345 bar before transmission by underground pipeline. If the compression is carried out adiabatically and reversibly, determine the compressor...
-
Ethyl triflate is much more reactive than ethyl mesylate toward nucleophiles in SN2 reactions. (a) Give the structures of all of the products formed when each compound reacts with potassium iodide in...
-
Facebook : Which is the local/domestic market chosen by the specific company? History relating to how and when it entered the global market? Assessing and analyzing the opportunities and risks for...
-
For this assignment, I want you to identify a career or field you may be considering entering after you leave the university. Define your chosen career, its scope, and where you might expect to work....
-
Why is it important to give a neutral presentation when the goal is to inform?
-
Read Book: Baumgardner, Jennifer and Amy Richards. Manifesta [10th Anniversary Edition]: Young Women, Feminism, and the Future Chapter 7 Then answer: Who is Katie Roiphe and why do the authors spend...
-
What type of communication goals are designed to make a communicator be perceived in a particular way? Explain them in detail
-
What does a manufacturing cycle efficiency (MCE) of less than 1 mean? How would you interpret an MCE of 0.40?
-
In Exercises delete part of the domain so that the function that remains is one-to-one. Find the inverse function of the remaining function and give the domain of the inverse function. f(x) = 16x4 -3...
-
Which will be the strongest oxidizing agent under standard conditions (that is, all activities = 1): HNO 2 , Se, UO 2 2+ , Cl 2 , H 2 SO 3 , or MnO 2 ?
-
What is the difference between E and E for a redox reaction? Which one runs down to 0 when the complete cell comes to equilibrium?
-
(a) Use the Nernst equation to write the spontaneous chemical reaction that occurs in the cell in Demonstration 13-1. (b) If you use your fingers as a salt bridge in Demonstration 13-1, will your...
-
1. Write an Arduino sketch to perform the following: Repeatedly apply 5 V to your output pin and hold it for a period of 10t, then apply 0 V and hold for 10t. Read the input voltage Vin and the...
-
All draughts are at the perpendiculars 120 m apart. 24. The following are the half ordinates in m of a vessel for which the water- lines are 1.75 m apart and ordinates 25 m apart. Determine its...
-
PROBLEM EXERCISE: 1. A power plant is said to have had a use factor of 48.5% and a capacity factor of 42.4%. How many hours did it operate during the year? (answer 7660 hrs) 2. A 50 000kw steam plant...

Study smarter with the SolutionInn App


