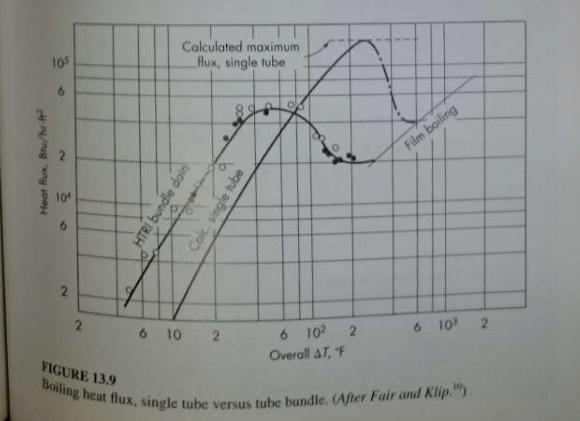
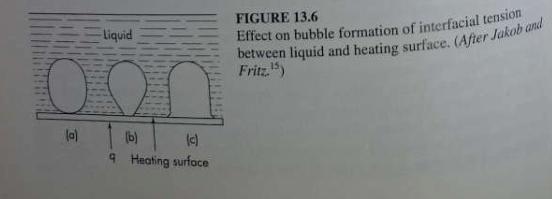
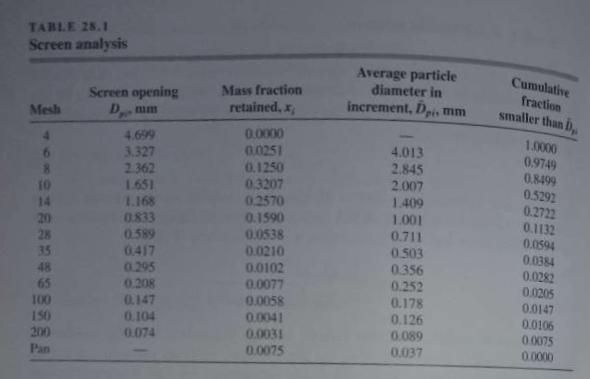
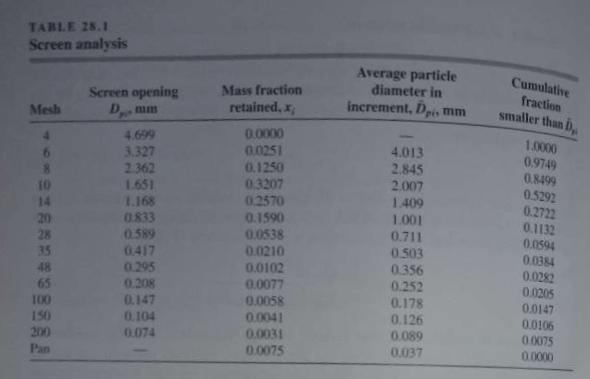
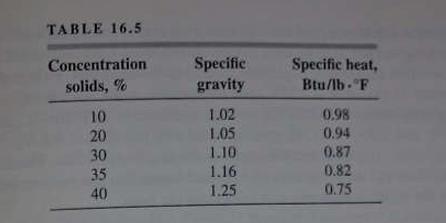
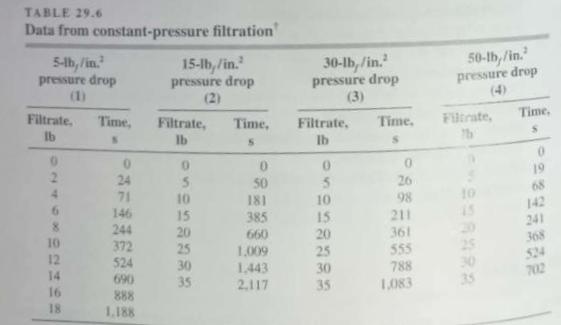
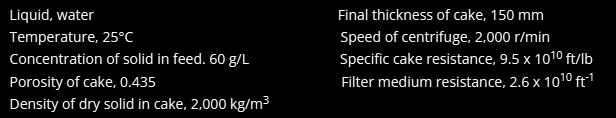
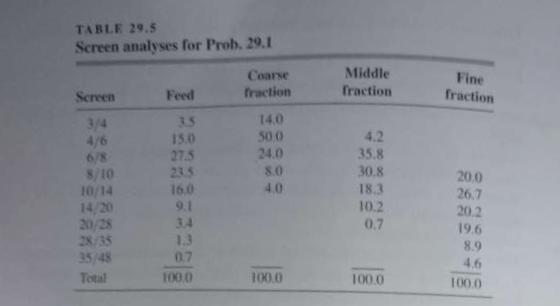
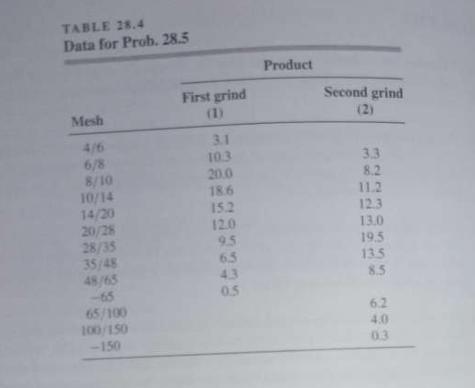
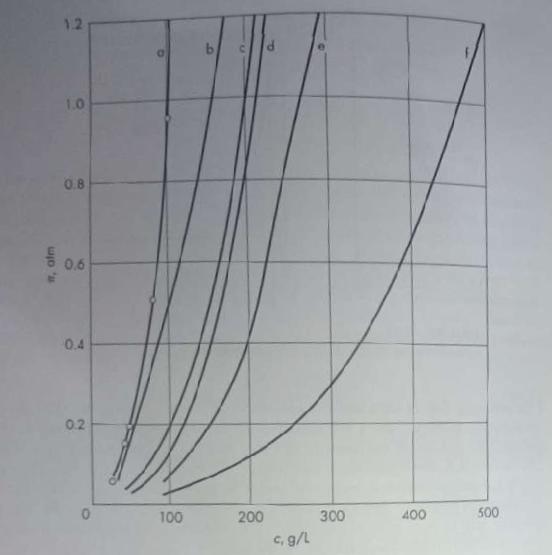
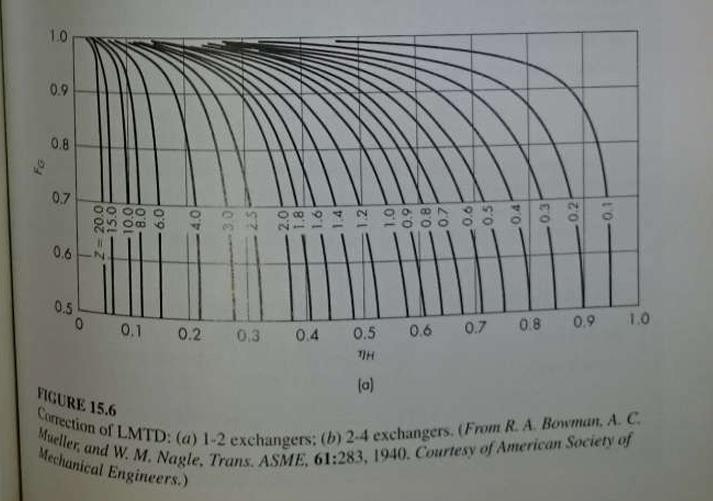
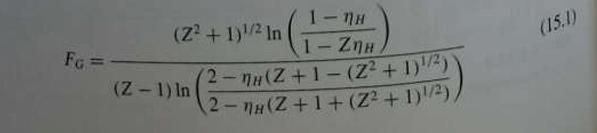Unit Operations Of Chemical Engineering 7th Edition Warren McCabe, Julian Smith, Peter Harriott - Solutions
Unlock the potential of your studies with our comprehensive resource hub dedicated to the "Unit Operations of Chemical Engineering 7th Edition" by Warren McCabe, Julian Smith, and Peter Harriott. Dive into our extensive collection of answers key and solution manuals, offering step-by-step answers and solved problems to enhance your understanding. Access chapter solutions, test bank insights, and instructor manuals, all designed to provide clear, concise questions and answers. Whether you’re searching for textbook solutions or a solutions PDF, our platform offers a seamless experience for online learning. Explore our free download options today!
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()