Using the values for the equilibrium constant for the ionization of water in Table 13.5-1, estimate the
Question:
Using the values for the equilibrium constant for the ionization of water in Table 13.5-1, estimate the standard-state heat of ionization of water as a function of temperature. Also, determine the pH of water at each of the temperatures in this table.
Table 13.5-1
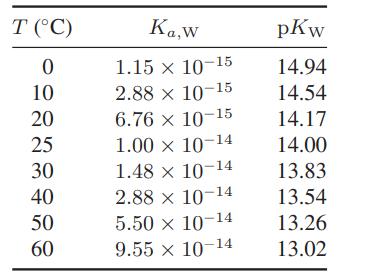
Transcribed Image Text:
T (°C) 0 10 20 25 30 40 50 60 Ka,w 1.15 x 10-15 2.88 x 10-15 6.76 × 10-15 1.00 x 10-14 1.48 × 10-14 x 2.88 x 10-14 5.50 x 10-14 9.55 x 10-14 pKw 14.94 14.54 14.17 14.00 13.83 13.54 13.26 13.02
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To estimate the standardstate heat of ionization of water as a function of temperature using the values given for the equilibrium constant Kaw from th...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The temperature distribution in a certain plane wall is where T1 and T2 are the temperatures on each side of the wall. If the thermal conductivity of the wall is constant and the wall thickness is L,...
-
The temperature of an ideal gas having constant specific heats is given as a function of specific entropy and specific volume as T(s,v) = Av1-k exp(s/cv) where A is a constant. For a reversible,...
-
For a more realistic estimate of the maximum coefficient of performance of a heat pump, assume that a heat pump takes in heat from outdoors at 10C below the ambient outdoor temperature, to account...
-
Determine which of the fundamental laws of algebra is demonstrated. 8(3 2) = 8(3) 8(2)
-
Divide the class into groups of six. Within each group, form pairs. One pair works Requirement 1(a); another pair works Requirement 1(b); and the remaining pair works Requirement 1(c). When the pairs...
-
Kailua Motors is a small manufacturer of specialty electric motors. The company employs 26 production workers and 7 administrative persons. The following procedures are used to process the companys...
-
James Lewis, a resident of Kentucky, sustained an injury while operating a Caterpillar bulldozer. He filed suit against Caterpillar, a company incorporated in Delaware but with its principal place of...
-
Matt Broderick Company began operations on January 2, 2013. It employs 9 individuals who work 8-hour days and are paid hourly. Each employee earns 10 paid vacation days and 6 paid sick days annually....
-
1. The rate at which a bean plant grows is given by a differentiable function R(t). measured in centimeters per day, where 0 st s 30. A graph of the function R is shown below along with a table of...
-
The behavior of hydrogen fluoride is unusual! For example, here are the critical properties of various hydrogen halides: has the lowest reported critical compressibility of any species. Experimental...
-
Derive Eqs. 13.5-8a and b. - MH+ = = 1 {{ MBOH + (MBOH) + Ka,w (M = 1) MOH-7 22 W 4Ka,w Y (M = 1) Kaw (M = 1) MBOHY (13.5-8a) Ka,w (M = 1) Ka, [MBON + (MBOH) + 4%, (M = 1) ] Y (13.5-8b)
-
Determine the clamping force on the board A if the screw of the C clamp is tightened with a twist M. The single square-threaded screw has a mean radius r, a lead h, and the coefficient of static...
-
CoffeeStop primarily sells coffee. It recently introduced a premium coffee-flavored liquor. Suppose the firm faces a tax rate of 22% and collects the following information. If it plans to finance 15%...
-
Boeing Corporation has just issued a callable (at par) three-year, 4.7% coupon bond with semiannual coupon payments. The bond can be called at par in two years or anytime thereafter on a coupon...
-
Your firm has tangible assets of $103.4 million. You are planning to acquire a firm that is half your firms size. You have bonds with a merger & acquisition covenant that requires the combined firm...
-
Despite the voracious habits of army ants, multiple species of invertebrates have managed to penetrate and exploit their societies. For example, the silverfish Malayatelura ponerophila is an insect...
-
Andyco, Inc., has the following balance sheet and an equity market-to-book ratio of 1.6. Assuming the market value of debt equals its book value, what weights should it use for its WACC calculation?...
-
Ellis issues 6.5%, five- year bonds dated January 1, 2013, with a $ 250,000 par value. The bonds pay interest on June 30 and December 31 and are issued at a price of $ 255,333. The annual market rate...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
Draw the shear and moment diagrams for the beam. 130 lb/ft 180 Ib ft . -9 ft- 4.5 ft
-
Draw the shear and moment diagrams for the beam. 20 kip 20 kip 4 kip/ft 111111111|| B -15 ft- 30 ft 15 ft
-
Draw the shear and moment diagrams for the beam. 4 kip/ft A 12 ft 12 ft
-
what is minimal data redundancy? could you please explain this ?
-
In your initial post to the discussion, answer the following questions in 2-3 paragraphs: What is data redundancy, and which characteristics of the file system can lead to it? What can a database...
-
If the following E-R model is transformed into a relational model below, does the relational model have the problem of data redundancy? If yes, describe what data is redundant in this relational...

Study smarter with the SolutionInn App


