Calculate the standard heat of the following reaction, given the enthalpies of formation: Standard enthalpies of formation
Question:
Calculate the standard heat of the following reaction, given the enthalpies of formation:
![]()
Standard enthalpies of formation kJ/mol
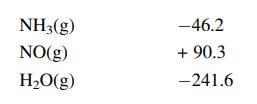
Transcribed Image Text:
4NH3(g) +50₂(g) →4NO(g) + 6H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The enthalpy of formation ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
You have been given the task of determining the standard heat of the reaction in which calcium chloride hexahydrate is formed from anhydrous calcium chloride: CaCl 2 (s) + 6 H 2 O (l) CaCl 2 6H 2...
-
Calcium chloride is a salt used in a number of food and medicinal applications and in brine for refrigeration systems. Its most distinctive property is its affinity for water: in its anhydrous form...
-
Trichloroethylene, a widely used degreasing solvent for machine parts, is produced in a two-step reaction sequence. Ethylene is first chlorinated to yield tetrachloroethane, which is...
-
1-Is it a good business strategy to have a day care center, Harley Davidson Motorcycle Dealership Sales and repair, Namaste Yoga studio, bookstore, temporary worker (day laborer) center, state prison...
-
The following paragraphs appeared in the New York Times on September 22, 1986. To keep the dollar from falling against the West German mark, the European central banks would have to sell marks and...
-
In the special report, "Bitter Pill: Why Medical Bills Are Killing Us" (TIME, Vol. 181, No. 8, 2013), S. Brill presented an in-depth investigation of hospital billing practices that reveals why U.S....
-
In Example 22.4, derive Eqs. (22.6) and (22.7). Then, graph the economic potential as a function of the reactor batch time for various values of the rate constant, \(k\), over the range \(0.4-0.6\)...
-
Mason owns Brickman, Inc., which specializes in laying brick patios, terraces, and walkways. Mason bids on a contract with State University to build several large terraces as well as the walkways...
-
Discuss the impact of non-ideal mixtures on distillation column design, focusing on vapor-liquid equilibrium (VLE) modeling and the use of activity coefficient-based models like UNIQUAC and NRTL
-
Hydrogen chloride gas, produced by burning chlorine with hydrogen, is required at a supply pressure of 600 kN/m 2 , gauge. The pressure can be achieved by either operating the burner under pressure...
-
Hydrogen chloride is produced by burning chlorine with an excess of hydrogen. The reaction is highly exothermic and reaches equilibrium very rapidly. The equilibrium mixture contains approximately 4...
-
What is cardiac output, and how is it calculated?
-
Your client want to have $168,313 in 20 years, how much money should he put in a savings account today? Assume that the savings account pays you 5.5 percent and it is compounded annually.
-
How Dominos Rose to the Top Problems/Issues; Possible Solutions; Recommend Solutions; and Expected Outcome?
-
What role does strategic foresight play in shaping strategic planning processes, particularly in anticipating and adapting to disruptive technological advancements, regulatory shifts, and...
-
Can you elaborate on the iterative nature of your strategic planning process, highlighting mechanisms for continuous learning, adaptation, and refinement in response to evolving environmental...
-
As a standard practice, the company does not recognize a provision for a guarantee of good operation of the products sold. Based on past statistics, the cost of repairing damage of products sold in...
-
Refer to the preceding facts for Presss acquisition of Simon common stock. Press uses the simple equity method to account for its investment in Simon. On January 1, 2013, Press held merchandise...
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
Suggest suitable materials of construction for the following applications: 1. A 10,000 m 3 storage tank for toluene; 2. A 5:0 m 3 tank for storing a 30% w/w aqueous solution of sodium chloride; 3. A...
-
Choose a suitable material of construction for the following duties: 1. 98% w/w sulfuric acid at 708C; 2. 5% w/w sulfuric acid at 308C; 3. 30% w/w hydrochloric acid at 508C; 4. 5% aqueous sodium...
-
A pipeline constructed of carbon steel failed after 3 years of operation. On examination it was found that the wall thickness had been reduced by corrosion to about half the original value. The...
-
After being business for 8 years, a customer gets scalded when a server accidentally spills hot espresso on them. The customer successfully sues your BizCafe for $200,000. How would this be handled...
-
The current tourism industry booms in Australia had given the HR department at ANIBT Hotel the opportunity to employ a very diverse range of ethnic employees for all levels of the industry. This...
-
We have distinguished several concepts of consciousness -Creature Consciousness, Transitive Consciousness, and Phenomenal Consciousnessbeing the most important. Explain what Change Blindness,...

Study smarter with the SolutionInn App


