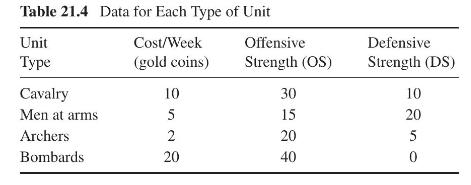
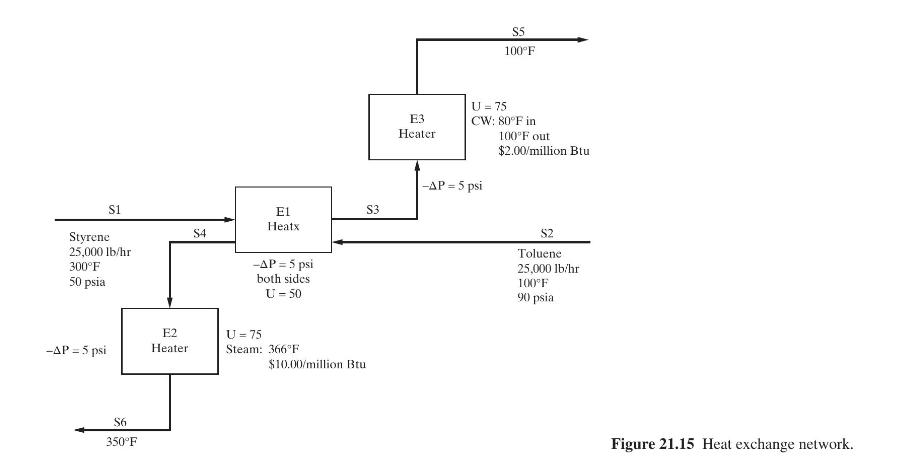
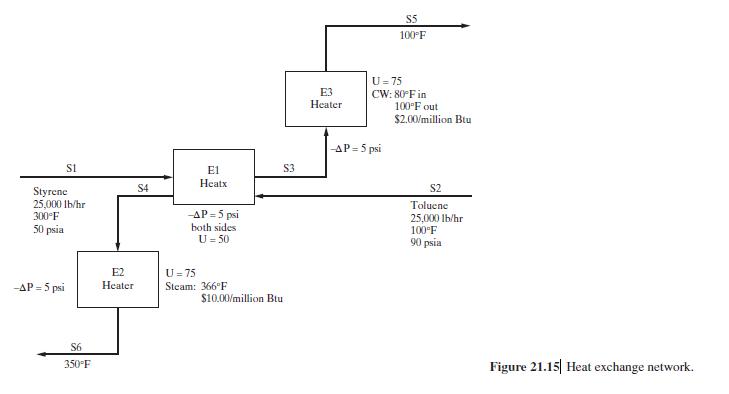
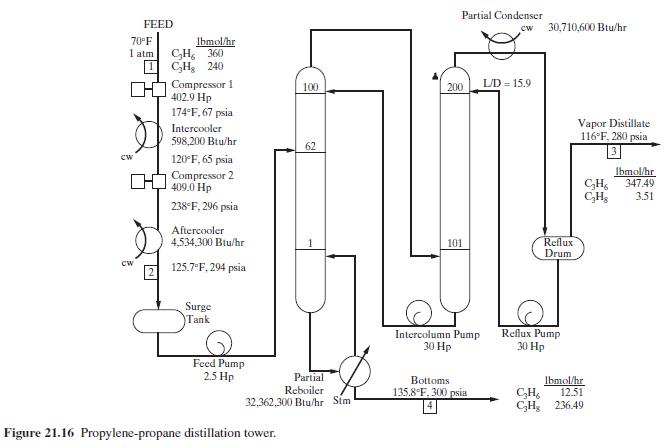
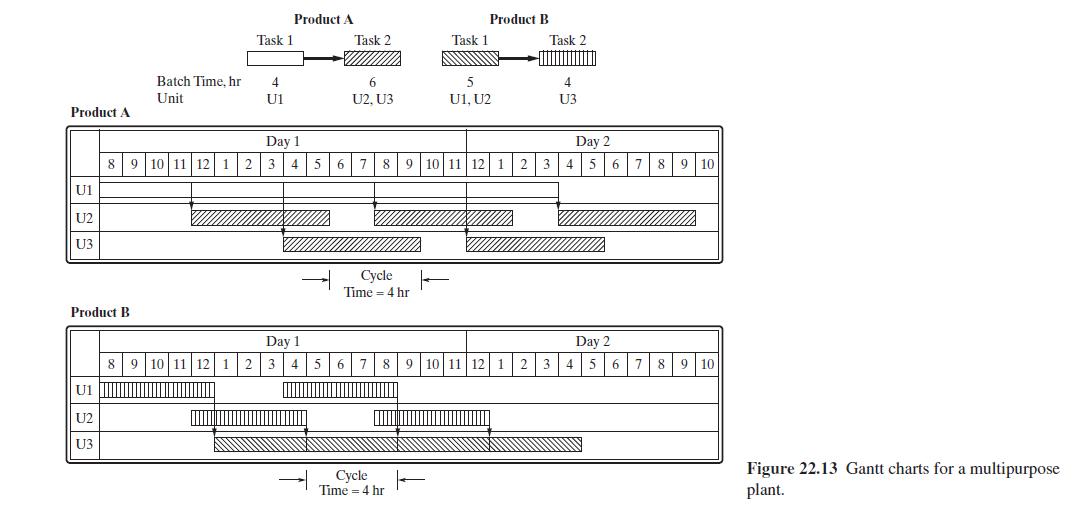
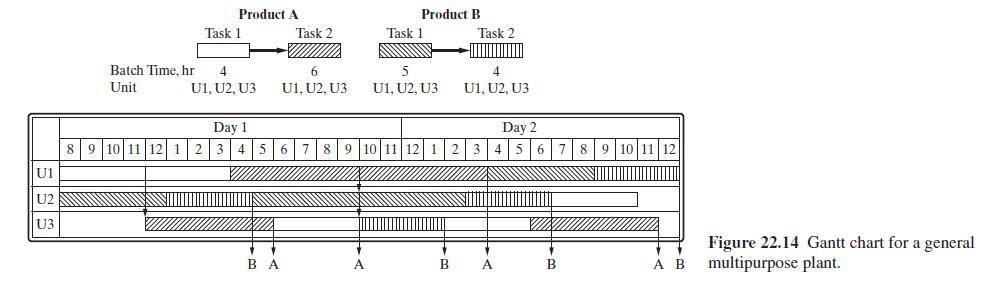
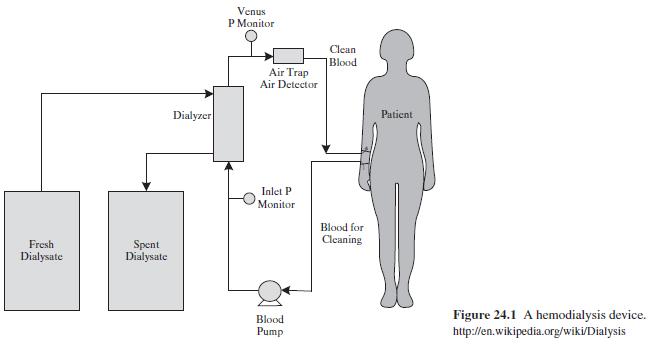
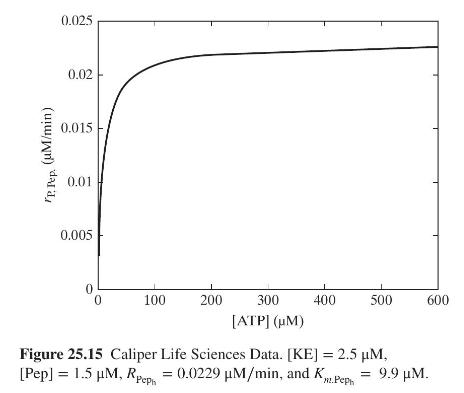
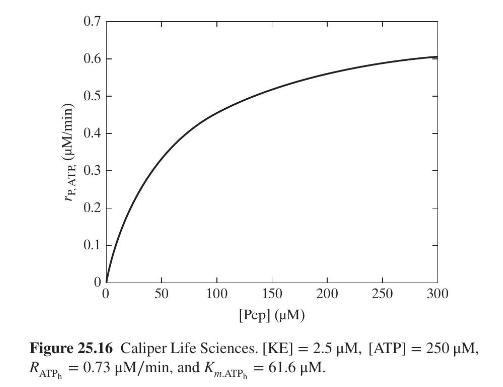
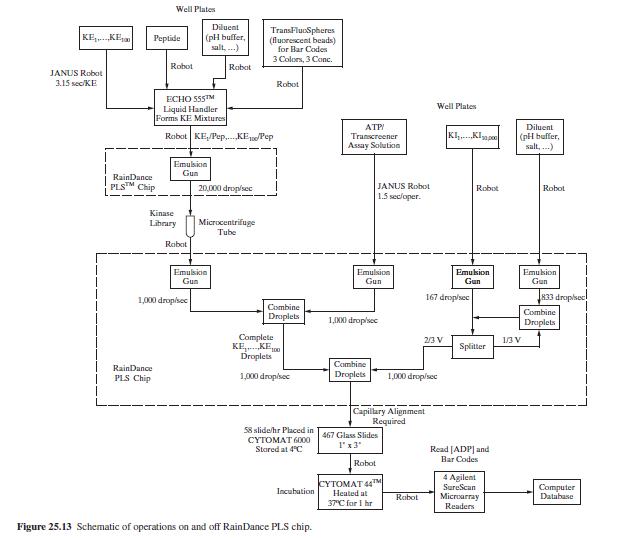
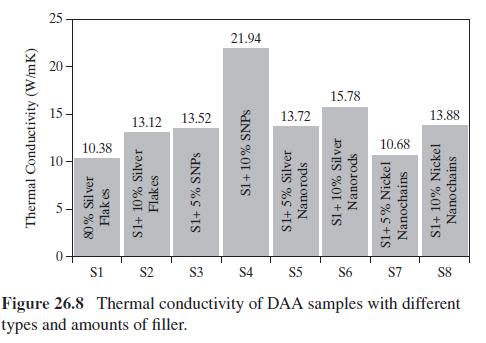
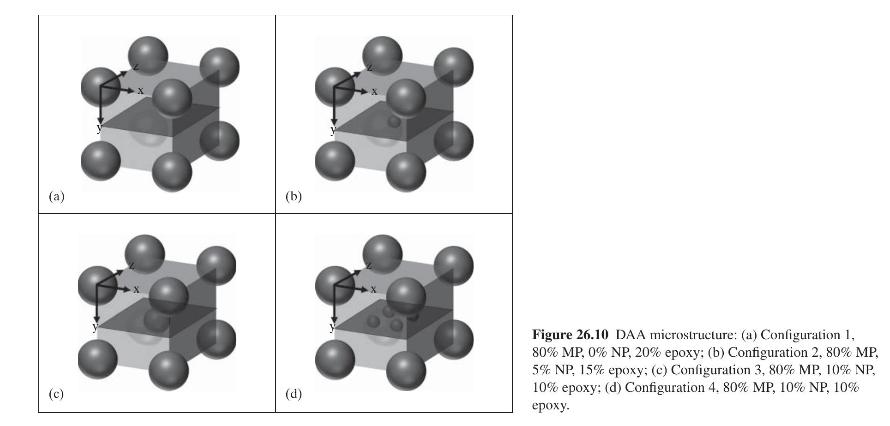
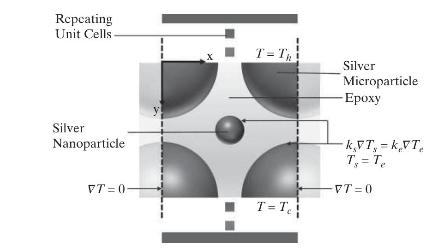
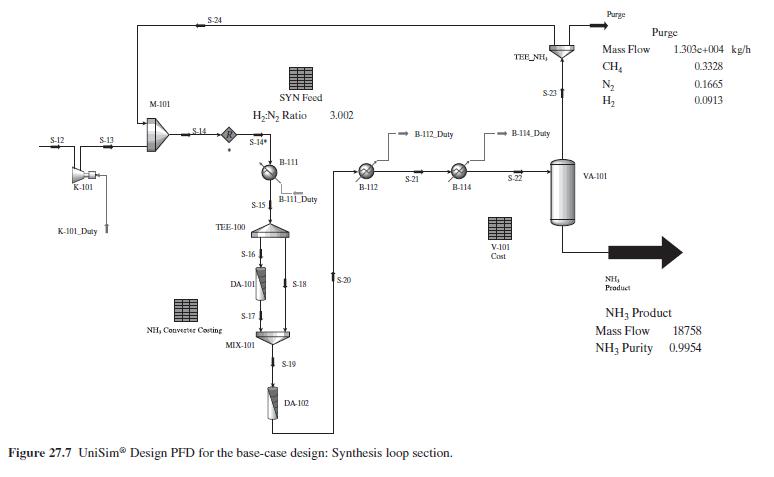
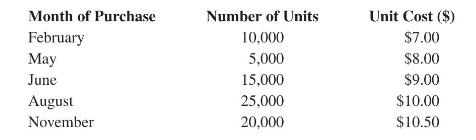
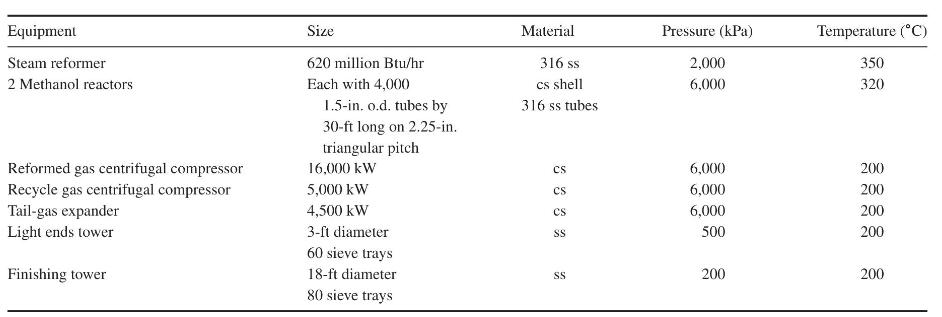
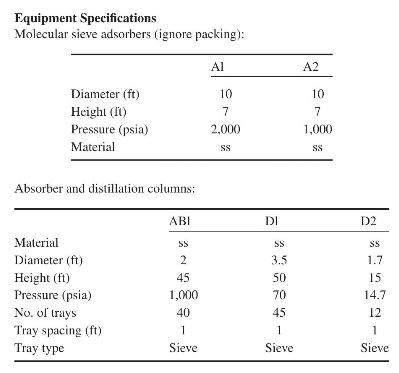
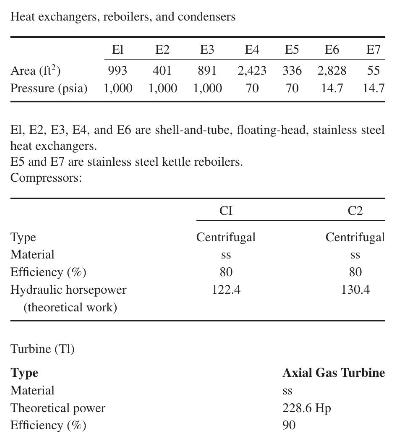
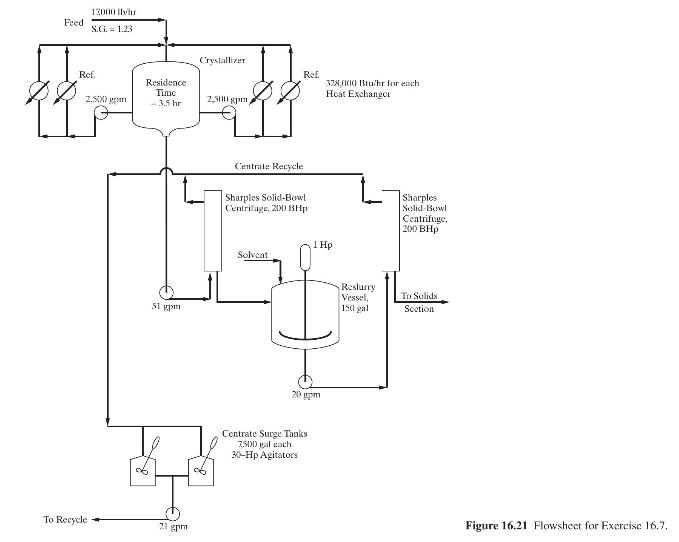
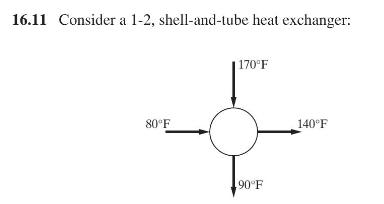
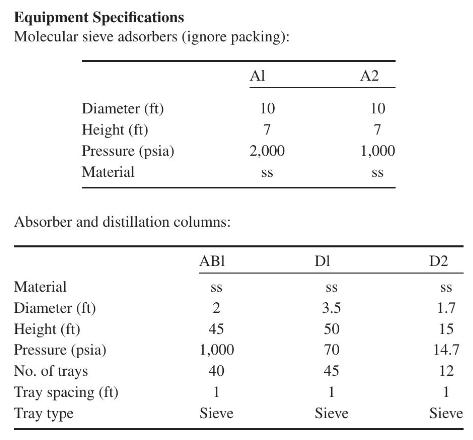
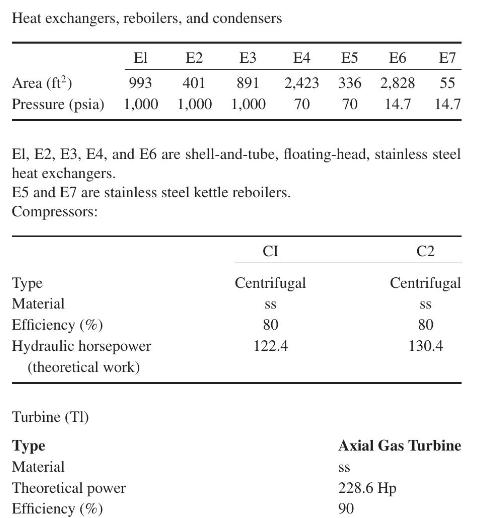
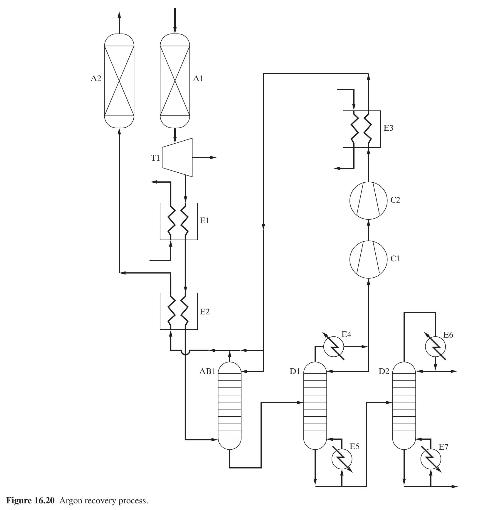
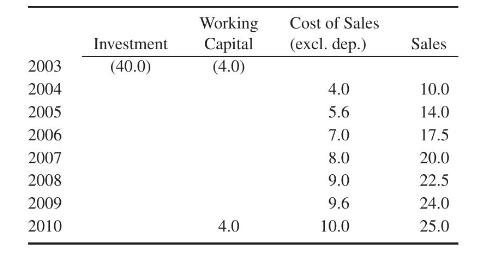
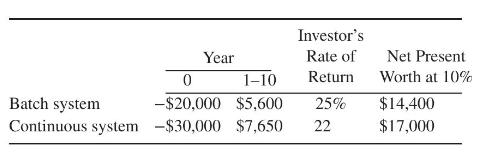
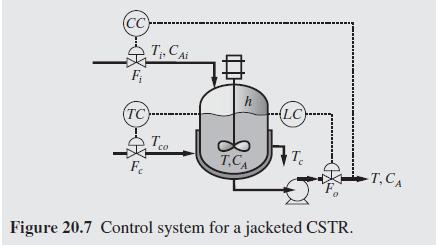
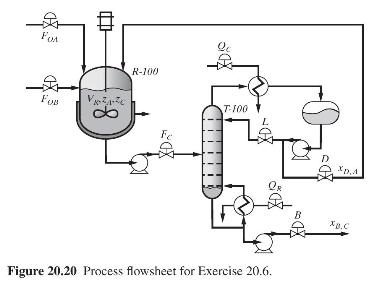
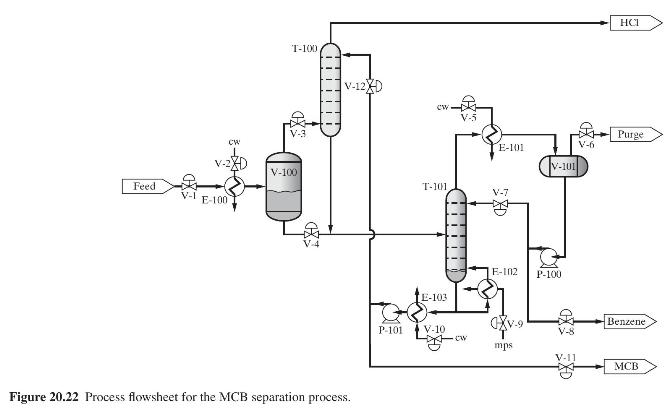
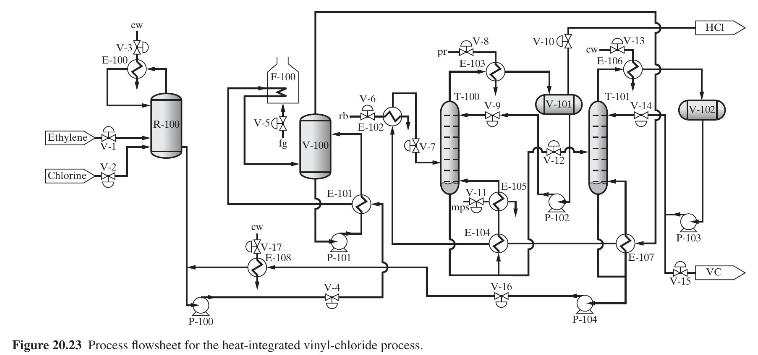
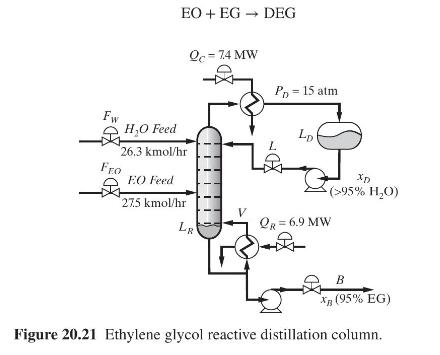
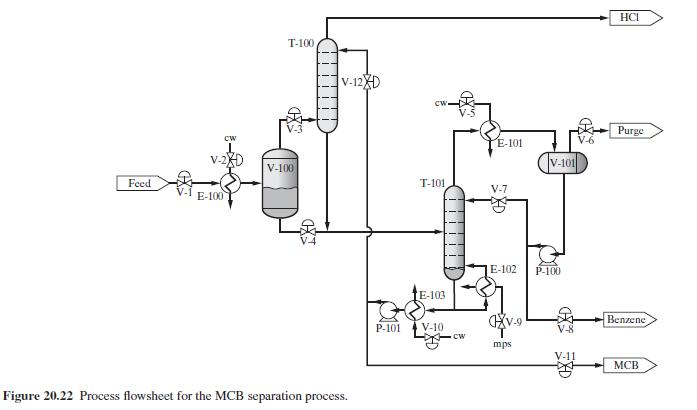
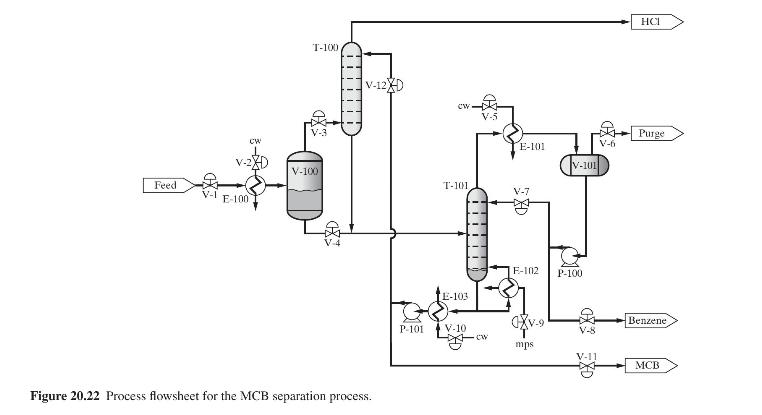
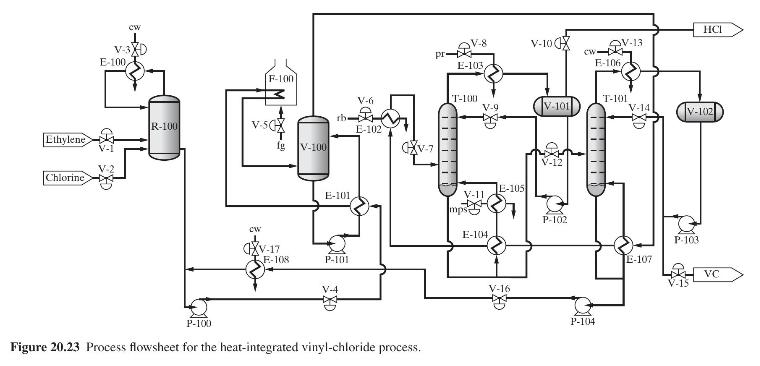
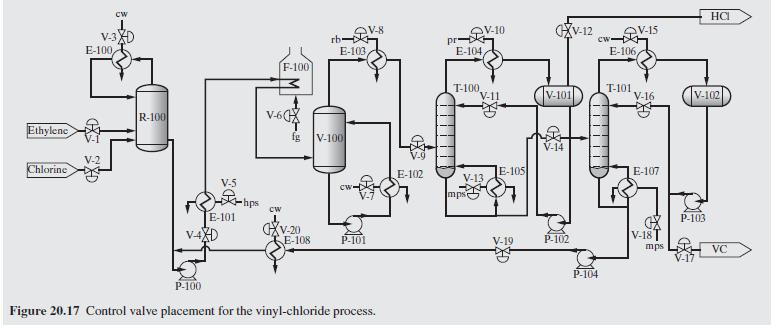
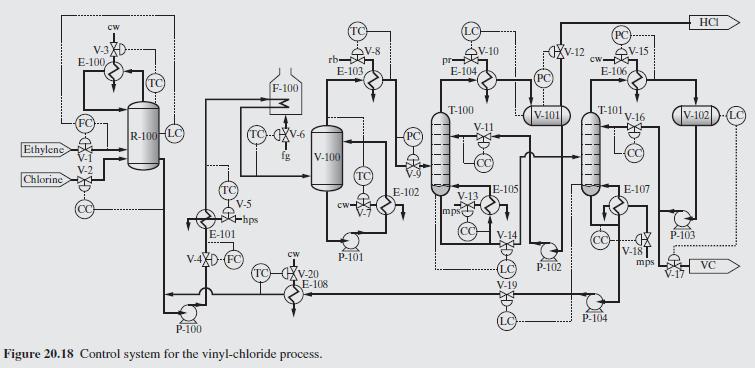
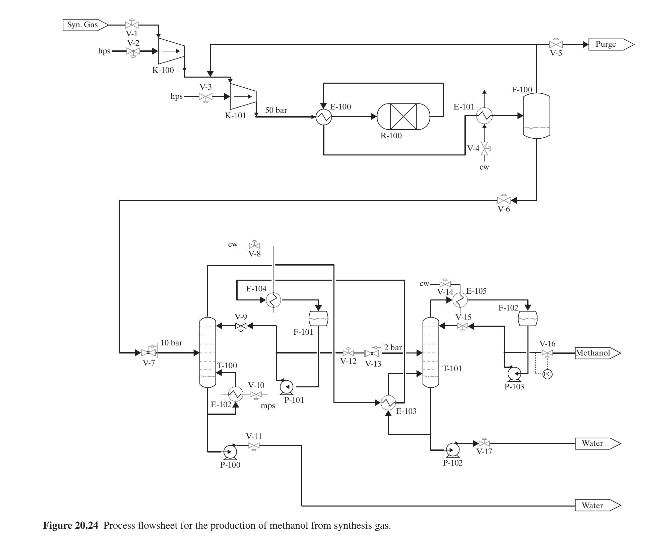
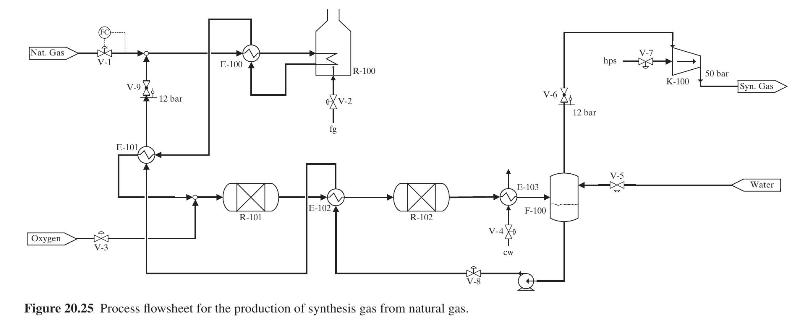
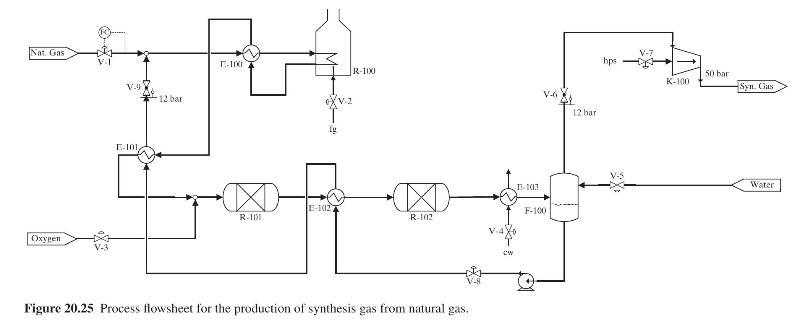
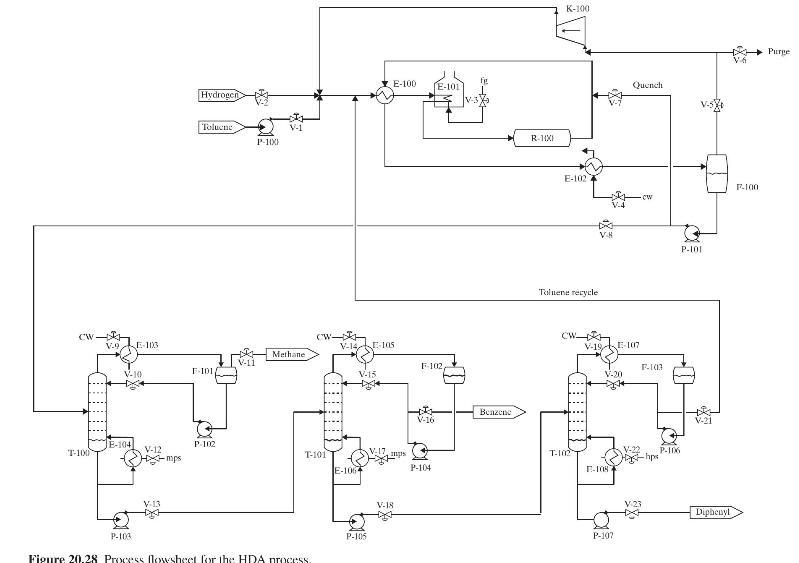
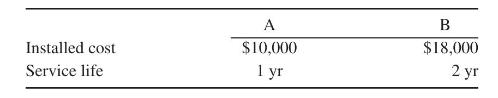Product And Process Design Principles Synthesis Analysis And Evaluation 4th Edition Warren D. Seider, Daniel R. Lewin, J. D. Seader, Soemantri Widagdo, Rafiqul Gani, Ka Ming Ng - Solutions
Discover comprehensive resources for "Product And Process Design Principles: Synthesis, Analysis, and Evaluation, 4th Edition" by Warren D. Seider and colleagues. Access a wide range of solved problems and step-by-step answers in our solution manual, ensuring you grasp each concept thoroughly. Whether you're looking for chapter solutions, an instructor manual, or a detailed test bank, our online platform offers convenient solutions in PDF format. Enjoy free downloads of textbook answers and enhance your learning experience with our expertly crafted answers key. Dive into questions and answers that simplify complex topics for optimal understanding.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()