Using Lydersens method, estimate the critical constants for isobutanol. Compare your values with those given in Appendix
Question:
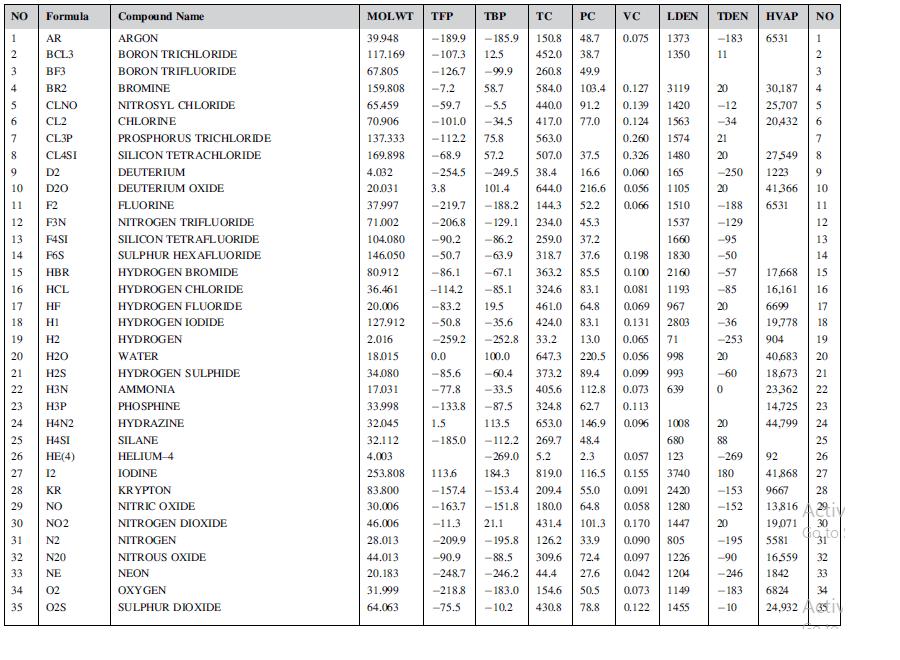
Using Lydersen’s method, estimate the critical constants for isobutanol. Compare your values with those given in Appendix C.
Data from appendix c
Transcribed Image Text:
NO 1 2 3 4 5 Formula AR BCL3 BF3 BR2 CLNO CL2 CL3P CLASI 15 16 8 9 D2 10 D20 11 F2 12 F3N 13 F4SI 14 F6S HBR HCL 17 HF 18 H1 19 H2 20 H2O 21 H2S 22 H3N 23 H3P 24 25 26 27 12 28 KR 29 NO 30 NO2 31 N2 32 N20 33 NE 34 02 35 02S H4N2 H4SI HE(4) Compound Name ARGON BORON TRICHLORIDE BORON TRIFLUORIDE BROMINE NITROSYL CHLORIDE CHLORINE PROSPHORUS TRICHLORIDE SILICON TETRACHLORIDE DEUTERIUM DEUTERIUM OXIDE FLUORINE NITROGEN TRIFLUORIDE SILICON TETRAFLUORIDE SULPHUR HEXAFLUORIDE HYDROGEN BROMIDE HYDROGEN CHLORIDE HYDROGEN FLUORIDE HYDROGEN IODIDE HYDROGEN WATER HYDROGEN SULPHIDE AMMONIA PHOSPHINE HYDRAZINE SILANE HELIUM-4 IODINE KRYPTON NITRIC OXIDE NITROGEN DIOXIDE NITROGEN NITROUS OXIDE NEON OXYGEN SULPHUR DIOXIDE MOLWT TFP 39.948 117,169 67.805 159.808 65.459 70.906 137.333 169.898 VC LDEN TDEN 0.075 1373 -183 1350 11 TBP PC -189.9 -185.9 -107.3 12.5 - 126.7 -99.9 260.8 49.9 TC 150.8 452.0 48.7 38.7 0.066 1510 1537 1660 0.198 1830 2160 -7.2 58.7 584.0 103.4 0.127 3119 -59.7 -5.5 440,0 91.2 0.139 1420 -101.0 -34.5 417.0 77.0 0.124 1563 -1122 75.8 563.0 0.260 1574 -68.9 57.2 507.0 37.5 0.326 1480 4.032 -254.5 -249.5 38.4 16.6 0.060 165 20.031 3.8 101.4 644.0 216.6 0.056 1105 37.997 -219.7 -188.2 144.3 52.2 71.002 -206.8 -129.1 234.0 45.3 104.080 -90.2 -86.2 259.0 37.2 146.050 -50.7 -63.9 318.7 37.6 80.912 -86.1 -67.1 363.2 85.5 0.100 36.461 -114.2 -85.1 324.6 83.1 0.081 1193 20.006 -83.2 19.5 461.0 64.8 0.069 967 -50.8 -35.6 424.0 83.1 0.131 2803 -259.2 -252.8 33.2 13.0 0.065 71 0.0 100.0 647.3 220.5 0.056 998 -85.6 -60.4 373.2 89.4 0.099 993 -77.8 -33.5 405.6 112.8. 0.073 639 -133.8 -87.5 324.8 62.7 0.113 1.5 113.5 653.0 146.9 0.096 1008 -185.0 -112.2 269.7 48.4 680 -269.0 5.2 2.3 0.057 123 184.3 819.0 116.5 0.155 3740 -153.4 209.4 55.0 0.091 2420 -153 -151.8 180.0 64.8 0.058 1280 -152 431.4 101.3 0.170 1447 20 126.2 33.9 0.090 805 -195 309.6 72.4 0.097 1226 -90 44.4 27.6 0.042 1204 -246 154.6 50.5 0.073 1149 -183 430.8 78.8 0.122 1455 -10 127.912 2.016 18.015 34,080 17.031 33.998 32.045 32.112 20 88 -269 180 4.003 253.808 83.800 30.006 46.006 28.013 113.6 -157.4 -163.7 -11.3 21.1 -209.9 -195.8 -90.9 -88.5 -248.7 -246.2 -218.8 - 183.0 -75.5 - 10.2 44.013 20.183 31.999 64.063 20 -12 -34 21 20 -250 20 -188 -129 -95 -50 -57 -85 20 -36 -253 20 -60 0 HVAP NO 6531 30,187 25,707 5 20,432 6 7 8 9 10 11 27549 1223 41.366 6531 2 17,668 16,161 6699 19,778 904 40,683 18,673 23.362 14,725 44,799 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 92 41,868 27 28 9667 13,816 19,071 30 Go to 31 16 Ativ 5581 16559 32 1842 33 6824 34 24,932 asti
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Lydersens method is a technique used to estimate the critical constants critical temperature critica...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
Make a rough estimate of the viscosity of 2-butanol and aniline at their boiling points, using the modified Arrhenius equation. Compare your values with those given using the equation for viscosity...
-
For intrinsic semiconductors, the intrinsic carrier concentration ni depends on temperature as follows: or taking natural logarithms, Thus, a plot of ln ni versus 1/T (K)-1 should be linear and yield...
-
Calculate the percentages of -D-glucose and -D-glucose present at equilibrium from the specific rotations of -D-glucose, -D-glucose, and the equilibrium mixture. Compare your values with those given...
-
How are the interface and implementation sections of an Objective-C class specified?
-
In order to determine the mass moment of inertia of a flywheel of radius 1.5 ft, a 20-lb block is attached to a wire that is wrapped around the flywheel. The block is released and is observed to fall...
-
In Exercises 1 and 2, describe the error. 1. (In e)2 = 2(In e) = 2(1) = 2 2. log2 8 = log2 (4 + 4) = log2 4 + log2 4 = log2 22 + log2 22 =2 + 2 = 4
-
A sensitive instrument of mass \(100 \mathrm{~kg}\) is installed at a location that is subjected to harmonic motion with frequency \(20 \mathrm{~Hz}\) and acceleration \(0.5 \mathrm{~m} /...
-
Many accounting and accounting-related professionals are skilled in financial analysis, but most are not skilled in manufacturing. This is especially the case for process manufacturing environments...
-
You purchase a bottle of water after entering the security checkpoint in an airport. You begin your flight, open the bottle and drink of the water, reseal it, and then land. Describe what has...
-
For the compounds listed below, estimate the constants in the equation for ideal gas heat capacity, equation 3.19, using the method given in Section 8.9.2. 1. 3-methyl thiophene. 2. Nitrobenzene. 3....
-
Evaluate the Dow F & EI for the nitric acid plant illustrated in Chapter 4, Figure 4.2. Data from figure 4.2 Flows kg/h Pressures nominal Line no. Stream Component NH3 0 N NO NO HNO3 HO Total Press...
-
Evaluate the integral using FTC I. J3 x dx
-
After taking the first part of this financial accounting course, you excitedly tell your friend Jordan what you have learned. You tell him about assets, liabilities and net worth and how they...
-
assume that T is a linear transformation. Find the standard matrix of T.
-
compute discounts, evaluate how discounts are used to influence consumer purchases, and identify the best discount option when shopping. Calculating the example: Disount is: $29.99 x 15% = $4.50 Sale...
-
Let A ERmxn with m
-
Consider the DE = 2x(2000-x). This is a logistic DE. (a) If a(t) is a solution to this DE then what are the possible values for limitt-x(t). (b) If a solution r(t) to this DE also satisfies 0 < x(0)...
-
Determine the direction of pulley B in each pulley system. 1. 2. 3. 4. 5. 6. What size pulley should be placed on a countershaft turning 150 rpm to drive a grinder with a 12.0-cm pulley that is to...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
Referring to the description in Problem P3.16, if the viscosity of water is 0.01 poise, determine the value in terms of the units (a) Slug/(ft s) (b) kg/(m s). Problem P3.16 The property of a fluid...
-
The fuel efficiency of an aircrafts jet engines is described by the thrust-specific fuel consumption (TSFC). The TSFC measures the rate of fuel consumption (mass of fuel burned per unit time)...
-
An automobile engine is advertised as producing a peak power of 118 hp (at an engine speed of 4000 rpm) and a peak torque of 186 ft lb (at 2500 rpm). Express those performance ratings in the SI...
-
1. A long cylindrical rod (Fig. 1) of radius R = 2 cm and length L = 40 cm is made of a material that generates energy from within. The conductivity of the material is k = 15 W/m K. The temperature...
-
Using the given knee diagram, estimate the amount of torque needed to actuate the knee near 0 flexion (standing position) based on friction in the knee joint for a person of (a) 50 kg, (b) 60 kg, (c)...
-
A space is at a temperature of 75 F (24 C), and the relative humidity is 45 percent. Find (a) the partial pressures of the air and water vapor, (b) the vapor density, and (c) the humidity ratio of...

Study smarter with the SolutionInn App


