(a) From the graph of Maxwell distribution of speeds in Fig. 3D.6, estimate the most probable speed...
Question:
(a) From the graph of Maxwell distribution of speeds in Fig. 3D.6, estimate the most probable speed of the molecules for each molar mass.
(b) What happens to the fraction of molecules having a speed in the narrow range Δv centered on the most probable speed, vmp, as the molar mass of the gas increases (Eq. 8)?
Fig. 3D.6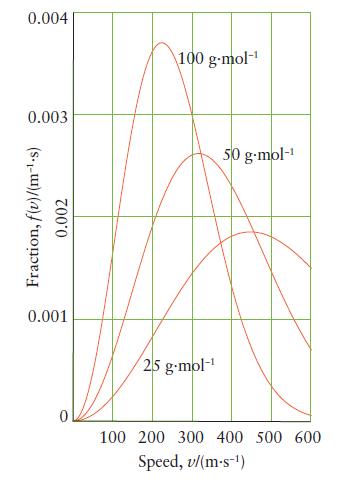
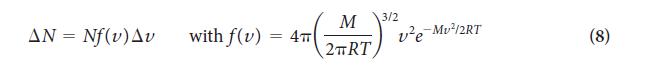
Transcribed Image Text:
0.004 0.003 Fraction, f(u)/(m-.s) 0.002 0.001 100 g-mol- 25 g-mol- 50 g.mol- 100 200 300 400 500 600 Speed, v/(m-s-)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The most probable speed for the molecule with molar mass of 25 g mol is at ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
(a) From the graph of Maxwell distribution of speeds in Fig. 3D.6, find the location that represents the most probable speed of the molecules at each temperature. (b) What happens to the fraction of...
-
The population of a culture of yeast cells is studied in the laboratory to see the effects of limited resources (food, space) on population growth. At 2-h intervals, the size of the population...
-
A vessel contains 1.00 X 104 oxygen molecules at 500 K. (a) Make an accurate graph of the MaxwellBoltzmann speed distribution function versus speed with points at speed intervals of 100 m/s. (b)...
-
Income statements for the current year and one year ago follow. Assume that all sales are on credit. For Year Ended December 31 Sales Cost of goods sold Other operating expenses Interest expense...
-
The tool is used to shut off gas valves that are difficult to access. If the force F is applied to the handle, determine the component of the moment created about the z axis of thevalve. F=1-601 +...
-
In Exercises find the integral by using the appropriate formula from Exercises. x sin x dx
-
A capacitor consisting of two concentric spheres and one consisting of two coaxial cylinders both have an inner radius of \(10.0 \mathrm{~mm}\) and an outer radius of \(30.0 \mathrm{~mm}\). If the...
-
The partnership of Larson, Norris, Spencer, and Harrison has decided to terminate operations and liquidate all business property. During this process, the partners expect to incur $8,000 in...
-
5. Identify the type of each of the following reactions stating reason for your answers: (a) Fe2O3 + 2Al 6. Al2O3 + 2Fe + heat (b) Pb(NO3)2 + 2KI PbI2 + 2KNO heat (c) ZnCOg ZnO + CO2 In the...
-
What percentage of space is occupied by close-packed cylinders of length l and radius r?
-
A sample of chlorine gas of volume 1.00 L at 1.00 atm and 298 K reacts completely with 1.00 L of nitrogen gas and 2.00 L of oxygen gas at the same temperature and pressure. There is a single gaseous...
-
The structures of [NMe 4 ][HF 2 ] and [NMe 4 ][H 2 F 3 ] have been determined by X-ray diffraction. The table on the next page shows selected structural data; all FHF angles are between 175 and 178....
-
Many airlines overbook flights. Is this efficient from an airlines point of view?
-
When the price of airline tickets falls, the supply falls and the demand increases. Using the economic ideas covered in this chapter, critically evaluate this statement.
-
How were airline ticket prices set during the period of regulation? From an economic point of view, what would be the predicted outcome of setting prices in this way?
-
Why is it important that an airline manager consider opportunity costs when making economic decisions regarding the airline?
-
Is price elasticity of demand for the airline industry in short-haul markets more or less than it is in long-haul markets? Why?
-
Classify each of the following occurrences as an incident or disaster. If an occurrence is a disaster, determine whether or not business continuity plans would be called into play. a. A hacker gets...
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
Which of the following sets of quantum numbers are not allowed? For each incorrect set, state why it is incorrect. a. n = 3, = 3, m = 0, ms = - 1/2 b. n = 4, = 3, m = 2, ms = - 1/2 c. n = 4, = 1,...
-
From the diagrams of 2p and 3p orbitals in Fig.19 and Fig.20, respectively, draw a rough graph of the square of the wave function for these orbitals in the direction of one of the lobes. Figure 12.19...
-
The wave function for the 2pz orbital in the hydrogen atom is Where a0 is the value for the radius of the first Bohr orbit in meters (5.29 Ã 10-11), Ï is Zr/a0, r is the value for the...
-
Discuss the impact of Australian government regulation on Venture Capital (a capital-raising mechanism) and compare the Australian regulatory environment to the United States. - Consider this from...
-
How retail loan consumers can use DTI information to help them shop for the appropriate lender? Comment on the methods and steps a consumer can take to reduce their DTI ratio. Are these methods...
-
Sanders Company has total assets of $ 3 8 9 million. Its total liabilities are $ 1 0 2 . 1 million, and its equity is $ 2 8 6 . 9 million. Calculate its debt ratio. ( Round your answer to 1 decimal...

Study smarter with the SolutionInn App


